The post Kidney Cancer Research Highlights from ASCO 2022 Annual Meeting first appeared on GUcancers.
]]>
.
Updated
Yasser Ged,1 and Nirmish Singla2,*
1) Department of Oncology, The Johns Hopkins University School of Medicine, Baltimore, MD, USA
2) Department of Urology, The James Buchanan Brady Urological Institute, The Johns Hopkins University School of Medicine, Baltimore, MD, USA
ABSTRACT
The 2022 American Society of Clinical Oncology (ASCO) annual meeting was held June 3-7, 2022, in Chicago, Illinois. This hybrid meeting gathered international cancer experts across multidisciplinary specialties and was held both virtually and in-person. Here, we highlight key kidney cancer research updates presented at the meeting. Slides from the meeting’s presentations are available on the ASCO meeting library website.
INTRODUCTION
Adjuvant Therapy Updates Locally advanced kidney cancer has traditionally been managed surgically alone1. However, approximately 30% of patients develop recurrent metastatic disease after surgical resection despite curative intent, and the optimal approaches to integrate surgery with systemic therapies in a neoadjuvant or adjuvant approach to reduce the risk of recurrence has been an area of active research.2 The U.S. Food and Drug Administration (FDA) has approved two adjuvant therapies in renal cell carcinoma (RCC) thus far, including sunitinib in 2017 and most recently pembrolizumab in 2021.3,4 The use of adjuvant sunitinib has been limited despite FDA approval because of its increased toxicity and lack of overall survival benefit.5 Pembrolizumab is the first approved adjuvant immunotherapy for clear cell RCC patients with intermediate-high or high risk of recurrence after nephrectomy based on the phase 3 doubleblind, multicenter, randomized KEYNOTE-564 study (NCT03142334).4
Updated analysis from KEYNOTE-564 was presented at the meeting evaluating the time to first subsequent drug treatment or any-cause death (TFST) and time from randomization to progression on next line of therapy or any-cause death (PFS2) after treatment with pembrolizumab or placebo in the study.6 Overall 67 patients (13.5%) in the pembrolizumab group and 99 patients (19.9%) in the placebo group received ≥1 line of subsequent anticancer drug therapy. A total of 108 PFS2 events were observed, 40 (8.1%; 12 death events and 28 progression events) in the pembrolizumab group and 68 (13.7%; 14 death events and 54 progression events) in the placebo group. PFS2 was also delayed with pembrolizumab compared with placebo (HR, 0.57; 95% CI, 0.39-0.85; medians not reached). The authors concluded that treatment with adjuvant pembrolizumab reduced risk for TFST and PFS2 compared with placebo. LITESPARK-022 (NCT05239728) is the next iteration of the KEYNOTE-564 study which is a phase 3 study designed to compare the efficacy and safety of belzutifan plus pembrolizumab with that of placebo plus pembrolizumab as adjuvant treatment for clear cell RCC after nephrectomy, and this study is currently actively enrolling.

.
Multiple adjuvant and neoadjuvant vascular endothelial growth factor tyrosine kinase inhibitors (VEGF-TKIs) studies in RCC were reported previously.5 To better understand the role of mammalian target of rapamycin (mTOR) inhibitors in the adjuvant setting, the Southwest Oncology Group (SWOG) launched the phase 3 study of everolimus in treating patients with kidney cancer who have undergone surgery (EVEREST) study (NCT01120249), which was reported at ASCO 2022.7 Individuals with clear or non-clear cell RCC immediately post-nephrectomy whose tumors show intermediate high-risk to high risk features were included in the study. Between 4/2011 and 9/2016, 1545 patients were randomized to e ither 1 2 m onths o f a djuvant e verolimus (n = 7 75) o r placebo (n = 7 70) including 83% w ith clear cell RCC and 17% with non-clear cell RCC. With a median follow-up of 76 months, the recurrence free survival was improved with everolimus compared to placebo (HR 0.85, 95% CI, 0.72 – 1.00; P (one sided) = 0.0246), narrowly missing the pre-specified, one-sided significance level of 0.022 which accounted for interim analyses, and the effect of everolimus was especially pronounced in patients with very high risk disease. Adverse events were consistent with safety profiles of everolimus, although there was a high discontinuation rate of everolimus in this population (47%).

.
First Line Metastatic Kidney Cancer Treatment Updates The first line treatment landscape of metastatic RCC has rapidly evolved in recent years.8 New updates on some of the registration first line metastatic RCC studies were presented during the meeting. The CheckMate 9ER trial is a phase 3 trial which compared nivolumab plus cabozantinib versus sunitinib in patients with untreated advanced clear cell RCC and demonstrated superior overall survival (OS), progression free survival (PFS) and objective responses of the nivolumab plus cabozantinib combination9. Updated analysis from the depth of response was presented at ASCO 2022.10 Patients’ responses were classified as complete response (CR) or partial response (PR) subdivided by a tumor reduction of ≥80%–<100% (PR1), ≥60%–<80% (PR2), or ≥30%–<60% (PR3). Overall, greater proportions of patients receiving nivolumab plus cabozantinib had deeper responses versus sunitinib (CR, PR1, PR2), and deeper responses with nivolumab plus cabozantinib were associated with improved 12-months PFS rate versus sunitinib for CR (94.9% vs 82.4%), PR1 (81.3% vs 37.5%), and PR2 (72.1% vs 53.2%).
.
Updates on health-related quality of life (HRQoL) from the CheckMate-214 phase 3 clinical trial, which compared nivolumab plus ipilimumab versus sunitinib in patients with untreated advanced clear cell RCC, were also presented during the meeting.11,12 As previously reported, nivolumab plus ipilimumab was associated with improved HRQoL compared to sunitinib. At ASCO 2022, the investigators reported on a post-hoc analysis on the prognostic ability of HRQoL to inform the risk of disease progression or death. The results of the analysis showed that higher (better) baseline scores were associated with significantly reduced risk of death (HR [95% CI] for FKSI- 19 Total Score and DRS score was 0.83 [0.80-0.87] and 0.80 [0.76-0.84], respectively). Furthermore, patients with improved/stable HRQoL had a 52% reduction in risk of death compared to patients who had worsened (HR 0.48 [95% CI: 0.39-0.59]).
.
Post-hoc exploratory analyses of PFS2 were conducted in the KEYNOTE 426 (phase 3 study comparing pembrolizuamb plus axitinib versus sunitinib in patients with untreated advanced clear cell RCC)13,14 and the CLEAR (phase 3 study comparing pembrolizumab plus lenvatinib versus sunitinib in patients with untreated advanced clear cell RCC)15,16 studies. Both analyses demonstrated prolongation of PFS2 in patients who received pembrolizumab plus axitinib in KEYNOTE 426 study and pembrolizumab plus lenvatinib in the CLEAR study. Novel Kidney Cancer Therapies Highlights Several exciting data were presented on novel therapies in RCC. Batiraxcept is a GAS6-AXL inhibitor, a pathway which is overexpressed in clear cell RCC.17 Interim results of a phase 1b study of batiraxcept plus cabozantinib 60 mg daily were presented at the meeting.18 A total of 26 patients were enrolled in the phase 1b study so far, and the recommended phase 2 dose of batiraxcept was identified as 15 mg/kg every 2 weeks. Encouraging early anti-tumor efficacy results of the combination were observed with an objective response rate of 67% and 6 months PFS of 79%. Hypoxia-inducible factor 2α (HIF-2α) is a key oncogenic driver in RCC.19 Belzutifan is a HIF-2α inhibitor which was recently approved by the FDA for patients with VHL syndrome and currently under investigation in sporadic RCC.20,21 LITESPARK-001 is a phase 1 study which was designed to evaluate belzutifan in heavily pretreated RCC and showed durable antitumor activity and an acceptable safety profile.21 An update of the clear cell RCC cohort in the study with more than 3 years of total followup was presented at the meeting.22 With extended followup of 41 months, the objective response rate was 25% with 80% disease control rate and median PFS of 14.5 months (95% CI, 7.3-22.1). Belzutifan monotherapy continued to show a high rate of disease control and durable responses in this heavily pre-treated population.
The CALYPSO study results were presented at the meeting as well.23 This is a randomized phase II study of durvalumab alone or with savolitinib or tremelimumab in previously treated advanced clear cell RCC. Savolitinib is a potent MET inhibitor with established dosing and activity in papillary RCC; however, its role in clear cell RCC is unclear.24 Between 2017 and 2021, 139 patients were randomized across the treatment arms. Savolitinib alone and in combination with duravlumab was associated with modest confirmed response rates (5% and 13%, respectively) compared to confirmed response rates of 10% for durvalumab and 28% for durvalumab plus tremelimumab. All regimens studied in the trial appeared to be safe and tolerable.
SUMMARY
In summary, ASCO 2022 was enriched with novel results and concepts continually expanding the field of kidney cancer research. Indeed, the data presented are both hypothesis-generating and practice-informing. Herein, we highlighted a snapshot of some of the oral presentations from the meeting in the kidney cancer space; however, there are considerably more exciting abstract and poster presentations that are available for review on the meeting’s website. In addition to the scientific content, ASCO 2022 also provided ample opportunities for networking and collaborations among the academic kidney cancer community, with the first in-person option since the beginning of the COVID-19 pandemic.
.
REFERENCES
1. Campbell SC, Clark PE, Chang SS, Karam JA, Souter L, Uzzo RG. Renal Mass and Localized Renal Cancer: Evaluation, Management, and Follow-Up: AUA Guideline: Part I. J Urol. 2021;206(2):199-208.
2. Apolo AB, Msaouel P, Niglio S, et al. Evolving Role of Adjuvant Systemic Therapy for Kidney and Urothelial Cancers. Am Soc Clin Oncol Educ Book. 2022;42:1-16.
3. Mejean A, Ravaud A, Thezenas S, et al. Sunitinib Alone or after Nephrectomy in Metastatic Renal-Cell Carcinoma. N Engl J Med. 2018;379(5):417-427.
4. Choueiri TK, Tomczak P, Park SH, et al. Adjuvant Pembrolizumab after Nephrectomy in Renal-Cell Carcinoma. N Engl J Med. 2021;385(8):683-694.
5. Gleeson JP, Motzer RJ, Lee CH. The current role for adjuvant and neoadjuvant therapy in renal cell cancer. Curr Opin Urol. 2019;29(6):636-642.
6. Choueiri TK, Tomczak P, Park SH, et al. Adjuvant pembrolizumab for postnephrectomy renal cell carcinoma (RCC): Expanded efficacy analyses from KEYNOTE-564. 2022;40(16_suppl):4512-4512.
7. Ryan CW, Tangen C, Heath EI, et al. EVEREST: Everolimus for renal cancer ensuing surgical therapy—A phase III study (SWOG S0931, NCT01120249). 2022;40(17_suppl):LBA4500-LBA4500.
8. McKay RR, Bosse D, Choueiri TK. Evolving Systemic Treatment Landscape for Patients With Advanced Renal Cell Carcinoma. J Clin Oncol. 2018:JCO2018790253.
9. Choueiri TK, Powles T, Burotto M, et al. Nivolumab plus Cabozantinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med. 2021;384(9):829-841.
10. Suárez C, Choueiri TK, Burotto M, et al. Association between depth of response (DepOR) and clinical outcomes: Exploratory analysis in patients with previously untreated advanced renal cell carcinoma (aRCC) in CheckMate 9ER. 2022;40(16_suppl):4501-4501.
11. Motzer RJ, Tannir NM, McDermott DF, et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N Engl J Med. 2018;378(14):1277-1290.
12. Cella D, Hamilton M, Blum SI, et al. The relationship between health-related quality of life (HRQoL) and clinical outcomes in patients with advanced renal cell carcinoma (aRCC) in CheckMate (CM) 214. 2022;40(16_suppl):4502-4502.
13. Rini BI, Plimack ER, Stus V, et al. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med. 2019;380(12):1116-1127.
14. Powles T, Plimack ER, Stus V, et al. Pembrolizumab (pembro) plus axitinib (axi) versus sunitinib as first-line therapy for advanced clear cell renal cell carcinoma (ccRCC): Analysis of progression after first subsequent therapy in KEYNOTE-426. 2022;40(16_suppl):4513-4513.
15. Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus Pembrolizumab or Everolimus for Advanced Renal Cell Carcinoma. N Engl J Med. 2021;384(14):1289-1300.
16. Voss MH, Powles T, McGregor BA, et al. Impact of subsequent therapies in patients (pts) with advanced renal cell carcinoma (aRCC) receiving lenvatinib plus pembrolizumab (LEN + PEMBRO) or sunitinib (SUN) in the CLEAR study. 2022;40(16_suppl):4514-4514.
17. Rankin EB, Fuh KC, Castellini L, et al. Direct regulation of GAS6/AXL signaling by HIF promotes renal metastasis through SRC and MET. Proc Natl Acad Sci U S A. 2014;111(37):13373-13378.
18. Shah NJ, Beckermann K, Vogelzang NJ, et al. A phase 1b/2 study of batiraxcept (AVB-S6-500) in combination with cabozantinib in patients with advanced or metastatic clear cell renal cell (ccRCC) carcinoma who have received front-line treatment (NCT04300140). 2022;40(16_suppl):4511-4511.
19. Choueiri TK, Kaelin WG, Jr. Targeting the HIF2-VEGF axis in renal cell carcinoma. Nat Med. 2020;26(10):1519-1530.20. Jonasch E, Donskov F, Iliopoulos O, et al. Belzutifan for Renal Cell Carcinoma in von Hippel- Lindau Disease. N Engl J Med. 2021;385(22):2036-2046.
21. Choueiri TK, Bauer TM, Papadopoulos KP, et al. Inhibition of hypoxia-inducible factor-2alpha in renal cell carcinoma with belzutifan: a phase 1 trial and biomarker analysis. Nat Med. 2021;27(5):802-805.
22. Jonasch E, Bauer TM, Papadopoulos KP, et al. Phase 1 LITESPARK-001 (MK-6482-001) study of belzutifan in advanced solid tumors: Update of the clear cell renal cell carcinoma (ccRCC) cohort with more than 3 years of total follow-up. 2022;40(16_suppl):4509-4509.
23. Powles T, Mendez-Vidal MJ, Rodriguez-Vida A, et al. CALYPSO: A three-arm randomized phase II study of durvalumab alone or with savolitinib or tremelimumab in previously treated advanced clear cell renal cancer. 2022;40(17_suppl):LBA4503-LBA4503.
24. Choueiri TK, Heng DYC, Lee JL, et al. Efficacy of Savolitinib vs Sunitinib in Patients With METDriven Papillary Renal Cell Carcinoma: The SAVOIR Phase 3 Randomized Clinical Trial. JAMA Oncol. 2020;6(8):1247-1255.
Correspondence to:
Nirmish Singla, MD, MSCS. Departments of Urology and Oncology, The James Buchanan Brady Urological Institute, The Johns Hopkins School of Medicine. Baltimore MD 21287.
EMAIL: nsingla2@jhmi.edu
The post Kidney Cancer Research Highlights from ASCO 2022 Annual Meeting first appeared on GUcancers.
]]>The post Comparison of Papillary Renal Cell Carcinoma Type 1 and Type 2: A Secondary Data Analysis first appeared on GUcancers.
]]>1. Clemson University, Hampton, GA 30228 USA.
2. School of Nursing, Clemson University, Clemson, SC 29634.
3. Mathematical and Statistical Sciences, Clemson University, Clemson, SC 29634.
4. Department of Public Health Sciences, Clemson University, Clemson, SC 29634
ABSTRACT
OBJECTIVE: The overall aim of this study was to determine if there are significant differences between type 1 and type 2 papillary renal cell carcinoma (PRCC) that can be utilized by healthcare providers. MATERIALS AND METHODS: This study performed a secondary data analysis using The Cancer Genome Atlas Kidney Renal Papillary Cell Carcinoma data to determine if there are clinically significant differences in survival, demographics (age, ethnicity, gender, and race), increased risk factors (body mass index [BMI] smoking history, neoplasm history, and malignancy history) and preferential genetic pathways between type 1 and type 2 PRCC tumors. RESULTS: Descriptive statistics were performed on a total of 156 cases to determine demographics, increased risk factors and genetic pathways. The hazard ratio, with type 1 as the reference group, was 2.459 (with 95% CI 0.9723, 6.217). Of the risk factor variables investigated, we found that smoking appeared to be associated with an increased risk of type 2 (OR 3.241 95% CI 1.066, 9.853). In the pathways analysis, we observed one significant difference between MAPK and PI3K, with the latter being significantly associated with type 2 (OR 4.968 95% CI 1.759, 14.031 Table 6). CONCLUSION: This study provides the framework for future more comprehensive research on the demographic, increased risk factor and genetic pathway differences between PRCC type 1 and type 2 tumors. Future investigations should include a more complete dataset with additional potential risk factors
INTRODUCTION
Renal cell carcinoma (RCC) is the 14th most common cancer worldwide and was the cause of 175,098 deaths in 20181. RCC consists of numerous subtypes including clear cell renal carcinoma, papillary renal cell carcinoma and most recently clear cell papillary renal cell carcinoma. Currently, papillary renal cell carcinoma (PRCC) is the second most common type of RCC, after clear cell renal cell carcinoma, comprising approximately 15-20% of all RCC cases2,3. PRCC is considered a heterogeneous disease consisting of two subtypes; type 1 and type 2. These subtypes are primarily distinguished by their histology and vary in prognosis, treatment and patient outcomes. Type 1 is histologically characterized by a single layer of cells with sparse basophilic cytoplasm and small oval shaped nuclei that are present in either the renal tubules or renal papillae. This type can be associated with both hereditary and sporadic PRCC.4,5 Conversely, type 2 tumors are histologically characterized by large pseudostratified cells with eosinophilic cytoplasm with large spherically shaped nuclei that are present in the renal papillae. These tumors can be associated with hereditary PRCC but are more often associated with the sporadic form of PRCC.6 Furthermore, research 6 has shown that patients with PRCC type 2 tumors are correlated with a higher rate of metastasis and have a lower overall survival rate compared with patients with type 1 tumors.7 The overall aim of this study was to determine if there are significant differences between type 1 and type 2 PRCC that can be utilized by healthcare p r o v i d e r s . Specifically, this study sought to determine if there are clinically s i g n i f i c a n t d i f f e r e n c e s in survival, demographics (age, ethnicity, gender, and race), increased risk factors (body mass index [BMI] smoking history, neoplasm history, and malignancy history) and preferential genetic pathways between type 1 and type 2 PRCC tumors.
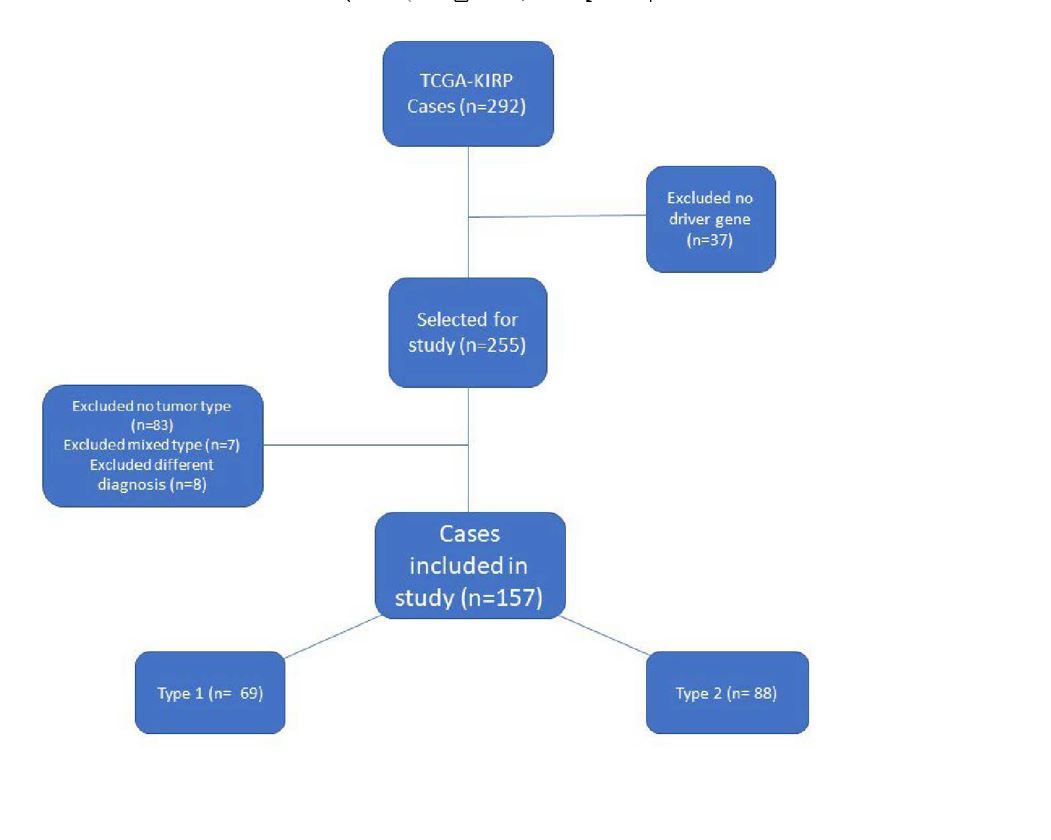
Table 1 | Descriptive Statistics for Demographic Factors
T h e epidemiology and risk factors for PRCC are largely based on the broader RCC. However, there are certain conditions that may increase an individual’s risk of developing PRCC. For instance, individuals with Hereditary Leiomyomatosis and Renal Cell Cancer (HLRCC) have a greater chance of developing PRCC type 2. There is some evidence that suggests individuals with renal insufficiencies have a greater risk of developing PRCC.8,9 Ethnicity is also linked to increased risk of developing RCC with African Americans having the highest incidence of RCCs. Sankin et al. (2011) found that African Americans had a four times greater incidence of PRCC as compared to non-African Americans.10,11 Research has demonstrated that malignant tumors utilize a wide variety of genetic alterations to modify the normal cell cycle in order to be able to divide and grow without restrictions. These modifications are accomplished by altering cell signaling pathways to promote cell growth, angiogenesis and obstruct apoptosis.12 Considering the heterogeneous nature of PRCC, there are numerous genetic alterations that occur within both type 1 and type 2 PRCC. Approximately 20% of hereditary type 1 tumors have been associated with variations in the protooncogene mesenchymal epithelial transition (MET). However, sporadic type 1 tumors have numerous genes associations as well as chromosomal abnormalities. Type 2 tumors have also been correlated with a large number of genetic and chromosomal alterations.4,13 Similarly, research has shown that renal cancers in general utilize several signaling pathways. The alteration of MET has been shown to activate the MAPK and PI3K pathways as well as other proteins involved with tumor growth.14 Gaps in research still exist for determining if there are pathway preferences between type 1 and type 2 PRCC tumors.
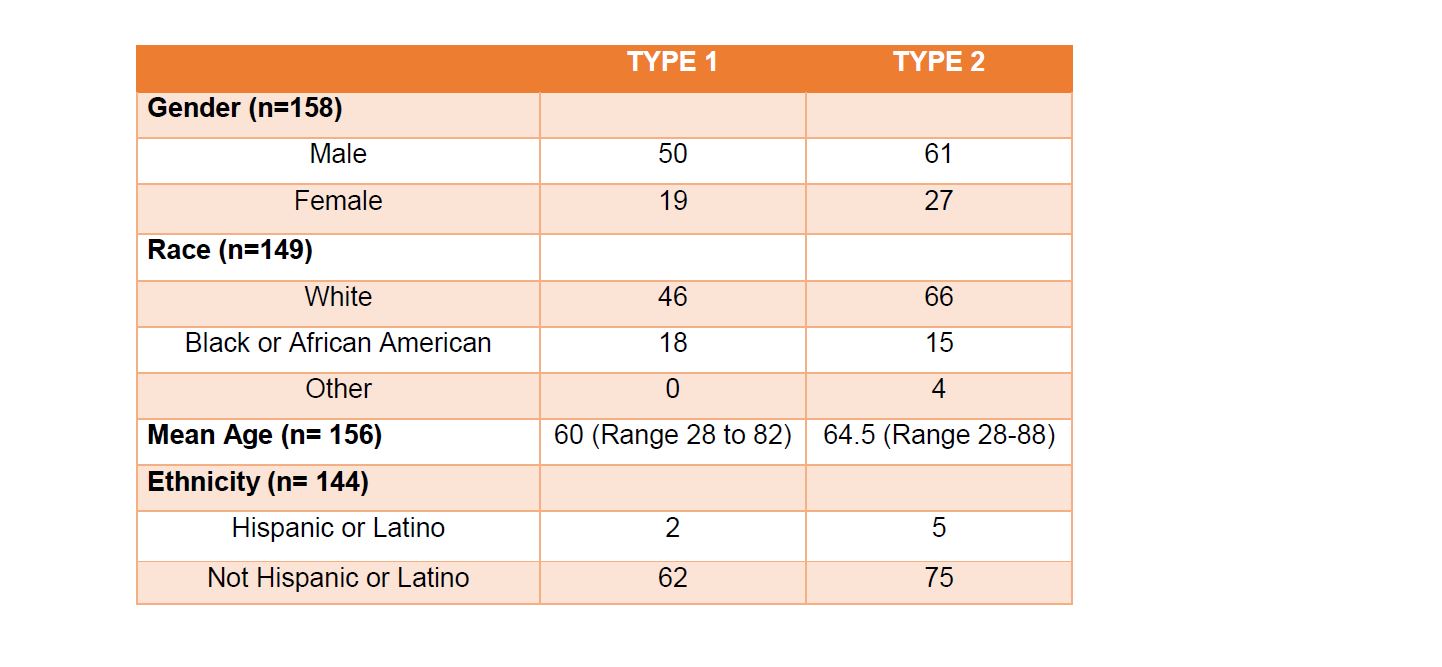
Table 1 | Descriptive Statistics for Demographic Factors
Most research on PRCC has either been umbrellaed under RCC or focused on developing a basic understanding of the disease with minimal attention to the differences between type 1 and type 2 PRCC tumors. Recently, Wong et al. (2019) investigated survival rates associated with type 1 and type 2 PRCC. The researchers found that type 2 PRCC was associated with a higher all-cause mortality rate as well as with worse reoccurrence rates as compared to type 1.7 As part of our research, we analyzed the allcause mortality for discrepancies in survival rates between type 1 and 2 PRCC. Next, we selected a demographic (baseline) model to identify a set of demographic variables that are likely to be associated with the different types of PRCC. Lastly, we investigated environmental and gene pathway associations with prevalence of the two types of PRCC.
METHODS
Sample
This study was a secondary data analysis using data from The Cancer Genome Atlas Kidney Renal Papillary Cell Carcinoma (TCGAKIRP). A review of the literature was conducted to determine the appropriate inclusion criteria which included: 1) PRCC tumors, 2) distinguishes between type 1 and type 2, 3) demographics data, gender, race, age and ethnicity, 4) clinical data, prognosis, treatment, preexisting conditions, 5) increased risk factors, smoking history, BMI, prior neoplasms and prior malignancies, and 6) genetic analysis of the tumors. A further review of the literature revealed that TCGA-KIRP is the most current and appropriate dataset to use for this secondary data analysis. The cBioPortal for cancer genomics (cBioPortal) was used in conjunction to analyze the TCGA-KIRP data.
TCGA-Kidney Renal Papillary Cell Carcinoma (KIRP) data was collected from 41 institutions from 1996 to 2013. The database adheres to a strict inclusion policy; TCGA tumors are untreated samples that were snap frozen. Each tumor sample has to have a matched normal sample from the same patient which generally comes in the form of the patient’s blood. The tumors and subsequent molecular data are cross referenced by Biospecimen Core Resource (BCR) to ensure validity. Furthermore, the BCR analyzes each sample for pathological quality control. This maintains that TCGA has a highquality tumor samples as well as consistent molecular data.15 Additionally, each sample was reviewed by a panel of six experienced pathologist to in order to be classified into type 1, type 2 or unclassified PRCC. Moreover, any samples that were preclassified were reassessed by the same panel to ensure proper classification.15 The cBioPortal is a resource that incorporates data from TCGA as well as actively curates data sets from the literature into a researchfriendly source. The cBioPortal separates PRCC genetic variations into categories such as copy number variations and mutations. Furthermore, the cBioPortal predetermines and denotes driver genes through specific algorithms.16 The cBioPortal allows the user to analyze specific genes, as opposed to TCGA, which only allows users to view the dataset as a whole and does not denote potential driver genes.16 Even though the cBioPortal contains the same data as TCGA, the cBioPortal was used to aid in the analysis of TCGA data.
Data extraction
Both databases showed the same cases which totaled 292. The first step in evaluating the dataset was determining the demographic and clinical data. TCGA contained a manifest of demographic, clinical, and environmental data. This manifest was downloaded and converted into an Excel file. Once retrieved, the dataset was reviewed and irrelevant data was removed; such data included serum levels, blood cell counts, IDH level, tumor laterality, lymph node data, tumor dimensions, treatment data, tissue collection data, sample weights, calcium levels, and vial numbers. Data categories that were redundant were also eliminated.
Next, the cBioPortal resource was used to determine pertinent genetic information related to PRCC. The first step was to download the copy number alteration (CNA) data from this resource. A total of 10,837 genes exhibited a copy number variation. Genes that were not considered to be driver genes according to the GISTIC algorithm were eliminated from the dataset. This elimination left a total of 426 driver genes with CNA. The driver genes were then put into the BCG query to determine how many cases included one or more of the driver CNA genes. A total of 193 of the cases (66%) contained one of the driver CNA genes. In order to increase the sample population, mutated driver genes (as determined by Mutsig) were added to the query bringing the total of genes to 517 and 255 (87%) cases. Thirty-six cases did not have an association with one of the 517 driver genes and were eliminated. The driver genes were divided into categories based on their cytoband for future reference.
The remaining 255 cases were reviewed to determine whether or not they were designated type 1 or type 2 PRCC. Out of the 255 cases, 115 cases had no designation in the type category. The pathology report of each of the 115 cases was reviewed to see if a pathologist had designated the tumor as either type 1 or type 2. Seven more cases were determined to be a mix of type 1 and type 2 histology and were also removed. Additionally, eight more cases were either mislabeled as PRCC or determined to favor a different cancer type per the reviewing pathologist. These eight cases did not include a TCGA addendum that disputed the cancer typing and therefore were removed from this dataset. (See Figure 1). At the conclusion of this analysis, 88 cases were designated as type 2, 69 cases were type 1, and 83 cases were undesignated. The 83 undesignated cases were subsequently removed from the dataset in order to preserve the validity and continuity of the data.
ANALYSIS
Descriptive Statistics and Survival Analysis
Descriptive statistics were utilized to determine demographics, increased risk factors and genetic pathways. The survival analysis was conducted for the TCGA-KIRP analytic file using R version 3.6.2. , the survival(v3.2-13) and the survminer (v0.4.9) packages.21-23 A cox-proportional hazard model was fitted on the overall survival times of 156 patients (1 had a survival time of 0 indicating that they were diagnosed post-mortem or there was an error in entry) to determine if there were evidence that survival rates differ between type 1 and 2 PRCC.
Logistic RegressionFor the next three phases of our statistical analysis, SASTM software, Version 9.4 of the SAS system for Windows was utilized. The demographic model selection included age at diagnosis, race, ethnicity and sex, as candidate descriptors relating to PRCC tumor type. The demographic model selection utilized forward selection with a relaxed p value (<0.1) to determine the appropriate variables to be included in the model. The selected demographic model included Age at Diagnosis (OR 1.045 95% CI 1.014, 1.078, Table 5) as well as 3 Category Race (White, Black or African American and Other) was used as the baseline model for the increased risk factor variables. Each increased risk factor variable; BMI, smoking status, prior neoplasms and prior malignancies, were added univariately to the demographic model controlling for age at diagnosis and race to identify associations.
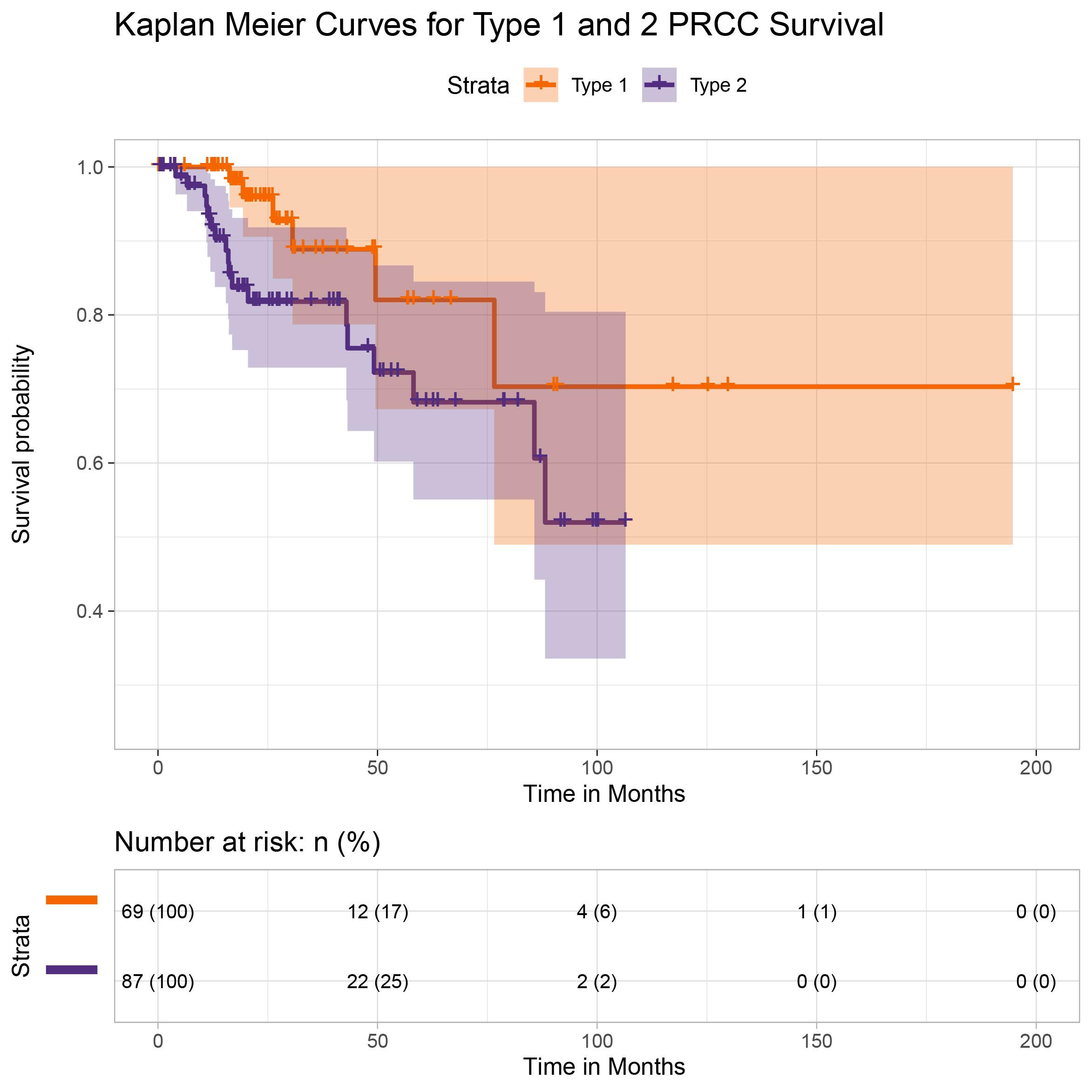
Figure 2 | Kaplan Meier curves for Type 1 and 2 PRCC survival
RESULTS
Descriptive StatisticsFor the 69 patients designated as type 1 tumors, 50 were male and 19 were female with a median age of 60 (range 28 to 82). In terms of race, 46 were white, 18 were black or African American, and 5 were unspecified. Ethnicity was reported as 62 non- Hispanic or Latino, 2 were Hispanic or Latino and 5 were unspecified.
For the 88 patients designated as type 2 tumors, 61 were male and 27 were female with a median age of 65 (range 28 to 88). In terms of race, 66 were white, 15 were black or African American, and 7 were unspecified. Ethnicity was reported as 75 were non-Hispanic or Latino, 5 were Hispanic or Latino and 8 were unspecified (Table 1). Due to the sparsity in the demographic factor levels, the following variable levels were collapsed; Asian and American Indian.
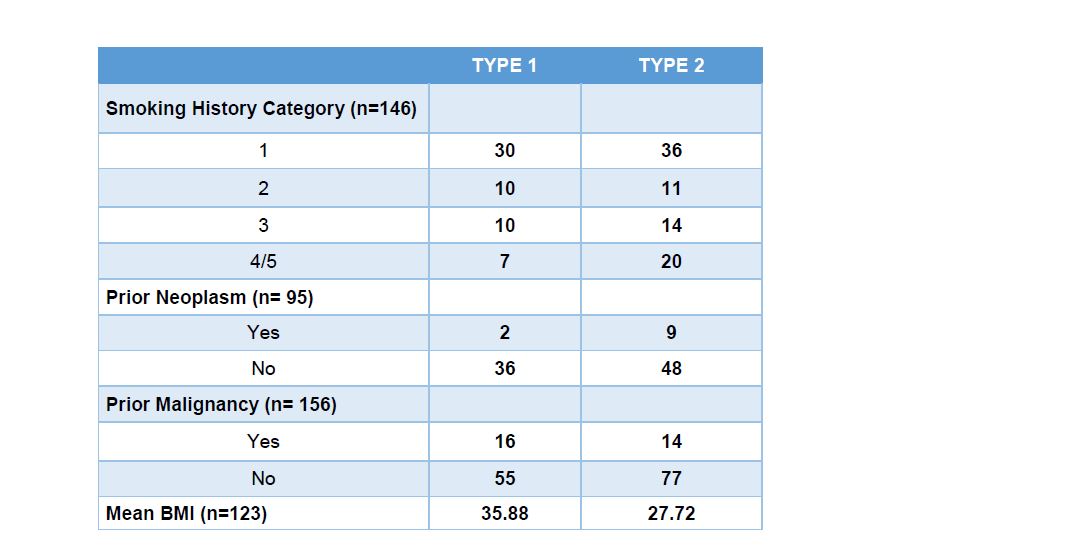
Table 2 | Descriptive Statistics for Increased Risk Factors
Smoking categories were defined as life-long non-smoker (1), current smoker (2), reformed smoker >15years (3), reformed smoker <15 years (4) and reformed smoker unknown length (5). Table 2 describes the smoking status of type 1 and type 2 PRCC tumors. Smoking categories 4 and 5 were collapsed together due to data sparsity in the increased risk factor variables. The existence of prior neoplasm was defined in the database as ‘yes’ or ‘no’. Two patients with type 1 PRCC had known prior neoplasm were as 9 patients with Type 2 reported prior neoplasm. Similarly, prior malignancies were also defined as ‘yes’ or ‘no’. Sixteen patients with type 1 reported prior malignancies and 14 patients with type 2 reported prior malignancies (Figure 2). The most common pathway in type 1 was the MAPK pathway and in type 2 was the PI3K pathway Table 3).
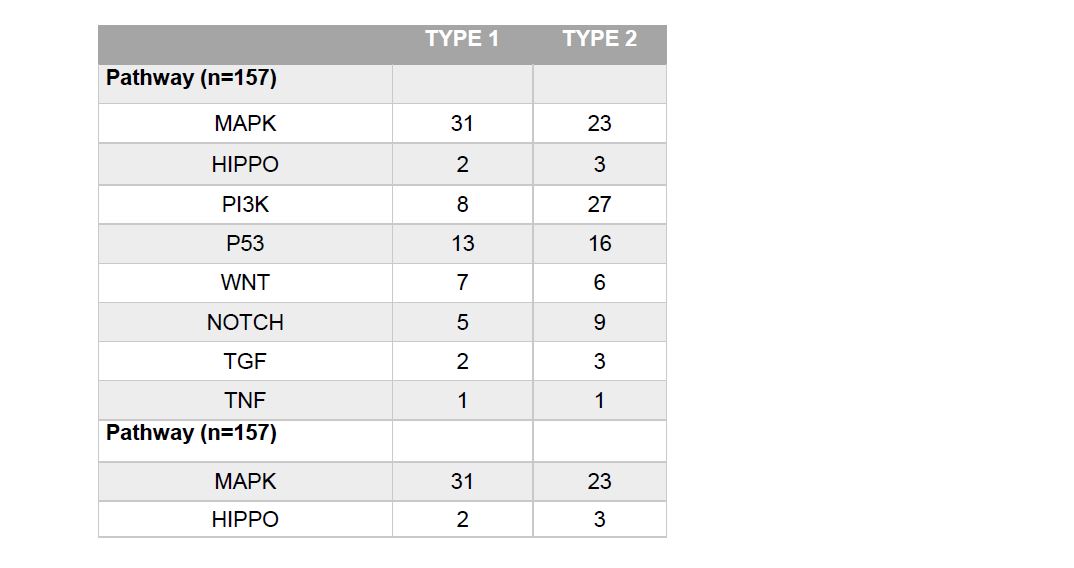
Table 4 | Demographics Model
Overall SurvivalThe hazard ratio, with type 1 as the reference group, was 2.459 (with 95% CI 0.9723, 6.217). This result did not provide sufficient evidence that the two types differ significantly in all-cause survival (α=.05). However, given the relatively small sample size and high rate of censoring, it is not surprising that our results do not provide as striking a contrast between the two as supported by Wong et al. (2019). (Censoring rates were 91.3% for Type 1 and 79.5% for type 2, respectively, which consequently prevents us from being able to report median survival without making parametric assumptions). Survival rates are illustrated via the Kaplan Meier curve included in Figure 2.
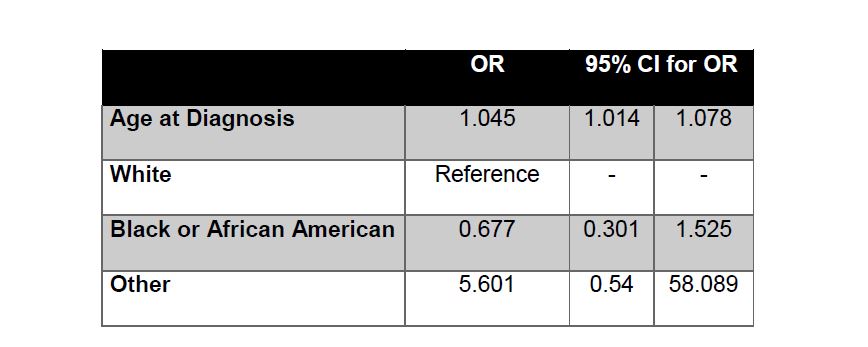
Table 4 | Increased Risk Factor Model
Logistic RegressionOdd ratios (OR) and confidence intervals (CI) are reported in Tables 5 and 6 for each variable in the increased risk factor and pathway analyses. Of the risk factor variables investigated, we found that smoking appeared to be associated with an increased risk of type 2. Specifically, being a reformed smoker of unknown length or less than 15 years, was positively associated with type 2 PRCC compared to lifelong non-smokers (OR 3.241 95% CI 1.066, 9.853 Table 5). None of the other increased risk factors had significant association with tumor type. In the pathways analysis, we observed one significant difference between MAPK and PI3K, with the latter being significantly associated with type 2 (OR 4.968 95% CI 1.759, 14.031 Table 6). All pairwise comparisons were made between pathways and the MAPK/PI3K comparison was the only one found to be significant. In all analyses, type 1 was used as the reference level for each model and the OR corresponds to odds of type 2 Vs 1.

Table 2 | Descriptive Statistics for Increased Risk Factors
DISCUSSION
It is important to note that current findings from the International Society of Urological Pathology (ISUP) suggests that the PRCC type 1 subtype is the most uniform morphologically, immunohistochemically, and in terms of molecular features. ISUP also suggests that PRCC type 2 is not a distinct neoplasm but rather a combination of multiple distinct neoplasms. As such, type 2 PRCC is a distinctly different disease as compared to type 1 and contains multiple clinically and molecularly heterogeneous subtypes.24 Additionally, the use of type 1 and type 2 terminology is evolving as PRCC becomes better understood. To the best of our knowledge, our study is the first to collectively examine the demographic, increased risk and pathway associations between type 1 and type 2 PRCC tumors. Furthermore, while our findings with respect to the survival analysis were not significant, it does provide marginal evidence to confirm the findings of Wong et al. (2019) in that survival rates for type 2 are shorter than those diagnosed with type 1. 7 While our analysis was limited by small sample size, certain variables were linked to increased probability of type 2 PRCC tumors. The age at diagnosis variable was considered significant with an older adult having increased risk of type 2. Our result is consistent with Wong et al. (2019) who reported a higher age at time of nephrectomy for patients with type 2 tumors as compared with type 1 tumors.7
Smoking was the only increased risk factor that was significant in determining the probability of having the type 2 tumor type versus type 1. Individuals who were reformed smokers of less than 15 years (as well as reformed smokers of unknown length) had a greater risk of developing a type 2 tumors as compared to lifelong non-smokers. Furthermore, type 2 PRCC tumors tend to be sporadic as compared to type 1, meaning that increased risk factors may have a greater impact on the development of type 2 tumors.6 However, further research needs to be conducted on the effects of smoking on the growth of specific tumor subtypes.
Although smoking was the only significant increased risk factor variable, further research should be conducted on a larger sample size with less missingness to better compare increased risk factors variables between tumor types. Specific focus should be put on prior neoplasms since they have been associated with a number of renal cell cancer syndromes that are considered to increase the risk of PRCC. For example, the most common renal cell cancer syndrome, von Hippel-Lindau syndrome, is characterized by benign tumor growths and has a 40% chance of developing renal cancer, including type 2 PRCC. Additionally, hereditary leiomyomatosis and renal cell cancer (HLRCC), is characterized by harmatomas with an increased risk of developing type 2 PRCC. 8,17 Considering the number of renal cell cancer syndromes that are both associated with an increased PRCC risk and are characterized by neoplasms; further research should be conducted to determine if prior neoplasms is a determining factor in PRCC subtype.
The findings in this study have potential implications for future treatment options. The higher rate of MAPK pathway in type 1 supports ongoing studies of the role of the MET gene in clinical trials. The MET gene codes for c-Met, a tyrosine kinase protein that is involved with the MAPK pathway. When c-Met binds to its ligand, HGF, a downstream cascade is started that leads to the activation of the MAPK pathway which promotes cell migration and tumor proliferation. 18 Seeing as 20% of type 1 tumors contain a MET mutation, it is not surprising that MAPK is the preferred pathway of type 1 tumors. Furthermore, the PI3K pathway was found to be significant in the probability of having a type 2 tumor as well as being the preferred pathway of type 2. The findings in this study support the ongoing efforts in determine drug treatment therapies that target the PI3K pathway. PI3K is comprised of lipid kinases that once activated, begin a downstream cascade that leads to cell growth and survival. PI3K pathway has a strong association with the inactivation of PTEN, which has been correlated poor patient outcomes.19,20
CONCLUSION
Despite the imperfect database this study found that there is a trend in the data that is clinically significant Furthermore, this study provides the framework for future more comprehensive research on the demographic, increased risk factor and genetic pathway differences between PRCC type 1 and type 2 tumors. Future investigations should include a more complete dataset with additional potential risk factors. Given the differences in survival rates, such investigations will provide clinicians a better understanding of tumor types allowing for quicker more accurate diagnosis and evidence-based treatment plans.
CONFLICT OF INTEREST
All authors listed on this study have no conflicts of interest that may be relevant to the contents of this manuscript.
FUNDING
None
ACKNOWLEDGMENTS
None
REFERENCES
Correspondence to: Melissa Paquin, PhD. 235 Galway Lane, Hampton, GA 30228 Email: mpaquin@clemson.edu
1. WHO. Cancer fact sheet. Retrieved from https://www.who.int/news-room/ fact-sheets/detail/cancer. 2020
2. Fernandes DS, Lopes JM. Pathology, therapy and prognosis of papillary renal carcinoma. Future Oncol. 2015;11(1):121- 132. doi:10.2217/fon.14.133
3. Steffens S, Janssen M, Roos FC, Becker F., Schumacher S, Seidel C, Wegener G, Thuroof T, Hofman R, Stockle M, et al. Incidence and long-term prognosis of papillary compared to clear cell renal cell carcinoma–a multicentre study. Eur J Cancer. 2012;48(15):2347-2352. doi:10.1016/j.ejca.2012.05.002
4. Marsaud A, Dadone B, Ambrosetti D, Baudoin C, Chamorey E, Rouleau E, Lefol C, Roussel JC, Fabas T, Crisofari G, et al. Dismantling papillary renal cell carcinoma classification: The heterogeneity of genetic profiles suggests several independent diseases. Genes Chromosomes Cancer. 2015;54(6):369- 382. doi:10.1002/gcc.22248
5. Procházková K, Mírka H, Trávníček I, Pitra T, Kolar J, Rousarova M, Hosek P, Bajcurova K, Ferda J, Staehler M, et al. Cystic Appearance on Imaging Methods (Bosniak III-IV) in Histologically Confirmed Papillary Renal Cell Carcinoma is Mainly Characteristic of Papillary Renal Cell Carcinoma Type 1 and Might Predict a Relatively Indolent Behavior of Papillary Renal Cell Carcinoma. Urol Int. 2018;101(4):409- 416. doi:10.1159/000492719
6. Yin X, Zhang T, Su X, Ji Y, Ye P, Fu H, Fan S, Shen Y, Gavine P, Gu Y. Relationships between Chromosome
7 Gain, MET Gene Copy Number Increase and MET Protein Overexpression in Chinese Papillary Renal Cell Carcinoma Patients. PLoS One. 2015;10(12):e0143468. Published 2015 Dec 4. doi:10.1371/journal. pone.01434687. Wong ECL, Di Lena R, Breau RH, Pouliot F, Finelli A, Lavallee L, So A, Tanguay S, Fairey S, Rendon R, et al. Morphologic subtyping as a prognostic predictor for survival in papillary renal cell carcinoma: Type 1 vs. type2. Urol Oncol. 2019;37(10):721-726. doi:10.1016/j.urolonc.2019.05.009
8. Paquin M, Fasolino T. Renal Cell Cancer Syndromes: Identification and Management of Patients and Families at Increased Risk. Clin J Oncol Nurs. 2020;24(4):356-359. doi:10.1188/20. CJON.356-359
9. Woldu SL, Weinberg AC, RoyChoudhury A, Chase H, Kalloo SD, McKiernan JM, DeCastro GJ. Renal insufficiency is associated with an increased risk of papillary renal cell carcinoma histology. Int Urol Nephrol. 2014;46(11):2127-2132. doi:10.1007/ s11255-014-0780-4
10. Hsieh JJ, Purdue MP, Signoretti S, et al. Renal cell carcinoma. Nat Rev Dis Primers. 2017;3:17009. Published 2017 Mar 9. doi:10.1038/nrdp.2017.9.11.Sankin A, Cohen J, Wang H, Macchia RJ, Karanikolas N. Rate of renal cell carcinoma subtypes in different races. Int Braz J Urol. 2011;37(1):29-34. doi:10.1590/s1677-55382011000100004
12. Sanchez-Vega F, Mina M, Armenia J, Chatila WK, Luna A, La KC, Dimitriaday S, Liu DL, Kantheti H, Saghafina S, et al. Oncogenic Signaling Pathways in The Cancer Genome Atlas. Cell. 2018;173(2):321-337.e10. doi:10.1016/j. cell.2018.03.035
13. Cancer Genome Atlas Research Network, Linehan WM, Spellman PT, Ricketts CJ, Creighton CJ, Fei SS, Davis C, Wheeler Da, Murray BA, Schmidt L, et al. Comprehensive Molecular Characterization of Papillary Renal-Cell Carcinoma. N Engl J Med. 2016;374(2):135-145. doi:10.1056/ NEJMoa1505917Luo G and Liu N. (2019). An integrative theory for cancer (Review). Int J Mol Med.;43(2):647-656. doi:10.3892/ijmm.2018.4004
14. Cojocaru E, Lozneanu L, Giuşcă SE, Căruntu ID, Danciu M. Renal carcinogenesis–insights into signaling pathways. Rom J Morphol Embryol. 2015;56(1):15-19.
15. The Cancer Genome Atlas. TCGAKIRP. Retrieved from https://portal.gdc. cancer.gov/projects/TCGA-KIRP.2020
16. cBioPortal. Papillary Renal Cell Carcinoma. Retrieved from https://www.cbioportal.org/study/ summary?id=kirp_tcga 2020
17. Modi PK, Singer EA. Improving our understanding of papillary renal cell carcinoma with integrative genomic analysis. Ann Transl Med. 2016;4(7):143. doi:10.21037/atm.2016.03.43
18. Zhang Y, Xia M, Jin K, Wang S, Wei H, Fan C, Wu Y, Li X, Li G, et al. Function of the c-Met receptor tyrosine kinase in carcinogenesis and associated therapeutic opportunities. Mol Cancer. 2018;17(1):45. Published 2018 Feb 19. doi:10.1186/s12943-018-0796-y
19. Yang J, Nie J, Ma X, Wei Y, Peng Y, Wei X. Targeting PI3K in cancer: mechanisms and advances in clinical trials. Mol Cancer. 2019;18(1):26. Published 2019 Feb 19. doi:10.1186/ s12943-019-0954-x
20. Bazzichetto C, Conciatori F, Pallocca M, Falcone I, Fanciulli M, Cognetti F, Miella M, Ciuffreda L. PTEN as a Prognostic/Predictive Biomarker in Cancer: An Unfulfilled Promise?. Cancers (Basel). 2019;11(4):435. Published 2019 Mar 28. doi:10.3390/ cancers11040435c
21. R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/. 2021.
22. Therneau T. A Package for Survival Analysis in R. R package version 3.3-1, https://CRAN.R-project.org/ package=survival. 2022.23. Alboukadel K., Kosinski M., and Biecek P. survminer: Drawing Survival Curves using ‘ggplot2’. R package version 0.4.9. https://CRAN.R-project. org/package=survminer. 2021
24. Williamson SR, Gill AJ, Argani P, Chen YB, Egevad L, Kristiansen G, Grignon D, and Hes O. Report From the International Society of Urological Pathology (ISUP) Consultation Conference on Molecular Pathology of Urogenital Cancers: III: Molecular Pathology of Kidney Cancer. Am J Surg Pathol. 2020;44(7):e47-e65. doi:10.1097/PAS.0000000000001476
The post Comparison of Papillary Renal Cell Carcinoma Type 1 and Type 2: A Secondary Data Analysis first appeared on GUcancers.
]]>The post Recent Advances in Tivozanib plus Nivolumab Combinatorial Strategies in Renal Cell Carcinoma first appeared on GUcancers.
]]>Robert A. Figlin, MD, FACP4
- Memorial Sloan Kettering Cancer Center, 1275 York Ave, New York, NY, 10065, USA.
- Lank Center for Genitourinary Oncology, Dana-Farber Cancer Institute, Boston, Massachusetts, USA
- Medical Oncology, Gustave Roussy, Villejuif, France.
- Cedars-Sinai Samuel Oschin Comprehensive Cancer Institute, Cedars-Sinai Health System, Los Angeles, CA

ABSTRACT
The treatment landscape of advanced renal cell carcinoma (aRCC) has witnessed significant benefits from the introduction of VEGF TKI/ICI (vascular endothelial growth factor receptor tyrosine kinase inhibitor/immune checkpoint inhibitor) combination in the first-line treatment. Such outcome benefits could extend to the relapsed/refractory setting with an effective, well-tolerated novel combination. Since the U.S. FDA approval of tivozanib monotherapy for the treatment of adult patients with relapsed or refractory advanced RCC following two or more prior systemic therapies,1 there is growing interest in exploring its full potential in combination with anti-PD-1 like ICI agents. In this roundtable discussion, internationally renowned cancer experts brainstorm the potential immunomodulatory capabilities of tivozanib plus nivolumab combination as first-liane and beyond settings in patients with metastatic RCC. The expert panel also explore potential data from previous and ongoing clinical trials and shared their perspectives about a tolerable safety profile and promising antitumor efficacy
Webinar Transcript
INTRODUCTION
In recent years, tyrosine kinase inhibitor/immune checkpoint inhibitor/ (ICI/TKI) combination regimens have emerged as novel treatment options for metastatic renal cell carcinoma (mRCC). However, it remains unclear how such combinations fit into the larger landscape of mRCC management, both in the first-line and beyond. Moreover, several clinical trials exploring such combinations have largely focused on the treatment-naïve population and their efficacy and toxicity beyond the first-line settings remain poorly defined.
Tivozanib, a highly selective and potent VEGF TKI, has demonstrated single-agent efficacy in advanced renal cell carcinoma.2,3 In addition, Tivozanib exhibits minimal off-target toxicities and a favorable adverse event (AE) profile.2-5 Based on these data, Tivozanib monotherapy was approved by the FDA on March 10th, 2021 for the treatment of adult patients with relapsed or refractory advanced RCC who have received 2 or more prior systemic therapies.1 As such, TKI/ICI combination regimens represent rationally designed novel therapeutic combinations built upon earlier work showing individual efficacy of each class of drugs in RCC. Tivozanib and nivolumab are ideal candidates for combination therapy owing to their efficacy, safety profile and a synergy between VEGFR and programmed death-1 (PD-1) inhibition in RCC.6 Tivozanib therapy facilitates immune-mediated responses through decrease in regulatory T cells (Tregs).7,8 The selectivity and favorable tolerability of the VEGFR TKI tivozanib9 may allow it to be used more readily as a combination therapy with an immune checkpoint inhibitor (ICI). Nivolumab an anti-PD-1 monoclonal antibody blocks the immune checkpoint protein PD-1 from interacting with its ligands programmed death ligands (PD-L1 and PD-L2). These mechanisms may act synergistically to potentially enhance the immune response that mediates antitumor activity.4

Following the FDA approval of tivozanib in renal cell carcinoma, tivozanib was explored in combination with the PD-1 inhibitor nivolumab in the Phase 1/2 TiNivo study,6 where it demonstrated favorable tolerability and prolonged PFS using the combination of tivozanib and nivolumab in both treatment naïve and previously treated patients with advanced RCC. Currently, the TiNivo-2 trial (NCT04987203)10 is exploring immunomodulatory effects and differentiated tolerability profile of the tivozanib plus nivolumab combination versus tivozanib monotherapy in a phase 3, randomized, controlled, multicenter, open-label study.10
The objective of this roundtable program is to further gain insights into the efficacy and tolerability of tivozanib plus nivolumab combination therapy for advance renal cell carcinoma patients. Also, leading oncology panelists share their insights that would enable clinicians to better understand full potential of tivozanib plus ICI combinations in a rapidly changing treatment paradigm of kidney cancers. The panel includes Drs. Robert Motzer, MD, Dr. Toni Choueiri and Dr. Laurence Albiges, MD and discussion was chaired by a renowned expert and our editor-in-chief, Robert A. Figlin, MD.
Below is an excerpt from the discussion edited for brevity and clarity.
ROUNDTABLE DISCUSSION
Dr. Figlin:
Welcome everybody. This is Robert Figlin, the Steven Spielberg Family Chair in Hematology-Oncology, Professor of Medicine and Biomedical Sciences, and Deputy Director of the Samuel Oschin Comprehensive Cancer Institute at Cedars-Sinai Medical Center in Los Angeles. On behalf of the kidney cancer journal, I am delighted today to have a roundtable with some distinguished investigators with great experience in the utility of tivozanib and its combination strategies, both in the clinical and research setting. So let me welcome Dr. Robert Motzer, Dr. Toni Choueiri and Dr. Laurence Albiges. Please introduce yourself to the audience.
Dr. Motzer:
I am an attending physician and Kidney Cancer Section Head in the Genitourinary Service, Department of Medicine, and Jack and Dorothy Byrne Chair in Clinical Oncology at Memorial Sloan Kettering Cancer Center in New York.
Dr. Choueiri:
I am the Director of the Lank Center for Genitourinary (GU) Oncology at Dana-Farber Cancer Institute (DFCI), co-leader of the Kidney Cancer Program at Dana-Farber/Harvard Cancer Center, and the Jerome and Nancy Kohlberg Chair and Professor of Medicine at Harvard Medical School.
Dr. Albiges:
I am Laurence Albiges a medical oncologist. I am head of Medical oncology department at the Gustave Roussy Institute in France.
Dr. Figlin:
You are distinguished group of panelists who join us today to discuss the roles of tivozanib. Dr. Motzer, let’s start with you. You are a pioneer in the development of tyrosine kinase inhibitors (TKI) and other potential therapeutics for the treatment of kidney cancers. Can you help us understand where tivozanib, a VEGF-TKI fits in that spectrum and the clinical trial that resulted in its approval in second line therapy patients that had received prior therapy?
Dr. Motzer:
Tivozanib is a potent and highly selective VEGF receptor tyrosine kinase inhibitor. It was developed at a very exciting time with many advances in our therapeutic armentarium between 2005 and 2012. At that time, there were several different VEGF TKIs including sunitinib, sorafenib and pazopanib which were studied in phase 3 trials and some of which were approved. Outstanding attributes of tivozanib in addition to efficacy are its tolerability and lack of off-target toxicities that had been seen with some other approved TKIs like sorafenib. Tivozanib was studiedin a randomized phase III TiVo-1 trial compared to sorafenib in treatment naïve or prior cytokine-treated subjects with metastatic RCC (mRCC). Based on the improved progression free survival (PFS), tivozanib was approved in Europe for first-line treatment of mRCC. However due to conflicting OS results, approval was put on hold in the United States. Given the potential efficacy in later lines of therapy, TIVO-3 was designed as an open-label phase 3, randomized, controlled, multicenter study to compare tivozanib to sorafenib in 350 subjects with refractory advanced renal cell carcinoma. The patients were heavily pretreated patients had two or three prior treatments, including a TKI and including some patients who have progressed on checkpoint inhibitors.
TIVO-3 met its primary endpoint by showing improvement in progression free survival compared to sorafenib among favorable and intermediate IMDC risk patients, indicating ongoing responsiveness to VEGFR inhibition . In the heavily pretreated population, tivozanib arm had a favorable safety profile which is a hallmark of tivozanib with fewer grade three or four adverse events, particularly those that are most troublesome to patients including hand foot syndrome and fatigue. The issue around overall survival was resolved, with both arms showing similar overall survival, and the tivozanib arm having superior response rate and progression free survival.
Dr. Figlin:
Toni, let me turn to you. You are a leader of combination therapies that have transformed the kidney cancer landscape in recent years. Can you help us understand the preclinical biological mechanism when combining a TKI with an IO? Do they become additive or synergistic? Are thereeffects on the tumor microenvironment that make those combinations better than either of those drugs alone?
Dr. Choueiri:
I can tell you that beyond seeing if there’s a synergy or additive effect, we are combining two drugs endowed with single agent activity of their own in frontline as well as refractory setting. Based on the clinical data, we cannot confirm that if there has been synergy or if each drug works on its own. Having said so, we know that in preclinical models, T cell mediated cancer cell killing, which is how immune checkpoint inhibitor largely works, may be enhanced through a reversal of VEGF mediated immunosuppression and other multiple steps in the immunity cycle. For example, the promotion of T cell priming and activation through the maturation of dendritic cells can happen by inhibiting VEGF. In addition, inhibiting VEGF can lead to the normalization of the tumor vasculature so that T cells could infiltrate the tumor better. VEGF inhibition can also lead to an immune permissive tumor microenvironment by decreasing the Myeloid-derived suppressor cells (MDSCs) and regulatory T (Treg) cells.
Dr. Figlin:
That’s quite a nice summary Toni! thank you. So Laurence let me let me turn to you. You’re the first author on the TiNivo-2 trial. It’s the Phase 1/2b trials that you’ve reported. So dive into that trial in terms of what you was the patient populations that you studied, the outcomes that you observed, and why that pivotal early trial has led to a larger trial in the phase III setting.
Dr. Albiges:
It’s actually building on two previous developments from Drs. Motzer and Choueiri based on the survival benefits obtained from single agent activities from both potent VEGF-TKI and immune checkpoint inhibitor and also as Dr. Choueiri highlighted that there is also an immunological standpoint as well. Based on such rational and also because of other combinations that were similarly developed, we launched an open-label, multicenter study of tivozanib in combination with nivolumab in patients with metastatic RCC (NCT03136627). In this study, patients initially received tivozanib 1.0 mg once daily for 21 days of treatment followed by 7 days off treatment. A standard dose of Nivolumab (240 mg) was administered as an infusion every 2 weeks starting on day 1 of cycle 1, and again on day 15. Overall, 25 patients have been treated as part of this phase I. patients with advanced RCC who received tivozanib plus nivolumab demonstrated a tolerable safety profile with minimal off-target effects when used as first-line and beyond treatment in patients with advanced RCC.
We assessed that 80% of our patient had some degree of remission, and the disease control rate was 96%. So overall, we were able to achieve a response rate of 56% in patients who received the tivozanib. Ultimately, the early signs of progression free survival was very enthusiastic with a median PFS of 18.9 months. So clearly, we observed a great signal of activity. What I can tell you from a clinical standpoint is that the tolerability was great, thanks to potency of tivozanib. The most frequent treatment-related toxicity was hypertension that was consistent with toxicity profiles of both drugs. Therefore it does require a close monitoring. However, as medical oncologists in the field of RCC had been exposed to hypertension for many years, hypertension was manageable from a clinical standpoint. The adverse events are comparable with other VEGFR TKI-PD-(L) and generally tolerable in combination setting.
Dr. Figlin:
Albiges Do you have any follow up data in terms of durable responses in those 25 patients that you’ve observed in your clinic?
Dr. Albiges:
The answer is yes. I could not speak for the entire study as it was a multi-centric study in France. However, I can tell you that I had seen very long, sustained response in my hospital. In some patients, we were able to discuss treatment discontinuation. So clearly what we’re seeing is a great disease control during the follow-up.
Dr. Figlin:
Before we turn to Toni and talk about the phase III trial. I think it’s important that we circle back and talk about quality of life benefits of tivozanib. Dr. Motzer, I know that you’ve reported on Quality of life data and the safety profile of tivozanib in the clinical setting. Please summarize some key objective data that you’ve reported on.
Dr. Motzer:
We performed quality of life analysis from the TiVo-1 trial, also accounting for efficacy and adverse event profile. In that direct comparison to serofenib, tivozanib was associated with a significant improvement in PFS and a favorable quality of life profile when administered to patients with metastatic RCC. In fact the toleratibility and safety profile is one of the greatest attributes of tivozanib. Tivozanib resulted in lower rates of certain side effects that are associated with the a decline in quality of life, including hand-foot skin reaction, rash anddiarrhea. There were fewer dose reductions and interruptions for tivozanib compared with sorafenib. In the other hand, tivozanib was characterized by higher rates of hypertension and dysphonia, but it was generally well tolerated.
Dr. Figlin:
Dr. Choueiri, let me get back to you. As a first author of TiNivo-2 study, please help us understand where you think that trial fits, what the goals and objectives are, and how that might offer our patients some continued immune modulation with positive outcomes in patients with mRCC?
Dr. Choueiri:
First, we were essentially looking into the unmet need in advanced RCC. One of the unmet needs is treatment for those patients whose tumors progressed after prior immune checkpoint inhibitors That’s why we launched TiNivo-2 study in this population based on the quite encouraging data from a phase 1/2b study that Dr. Albiges just mentioned. In TiNivo-2 trial, patients will be randomized to tivozanib monotherapy as the standard and the experimental arm have the combination of tivozanib plus Nivolumab. Patients should have been progressed at least one prior r line of therapy including an immune checkpoint inhibitor.. Subjects will be stratified by IMDC risk category and whether ICI was received in most recent line of treatment or not. Subjects will receive Tivozanib (1.34 mg orally once daily) for 21 consecutive days followed by 7 days off. In the combination arm, subjects will also receive Nivolumab 480mg intravenously every 4 weeks. The dose of tivozanib will be comparatively lower when delivered in combination with nivolumab as compared to tivozanib monotherapy. Ultimately, our goal here is to find a niche of completely unmet need and see if adding nivolumab on a backbone of VEGF TKI of tivozanib would result in improved outcomes in terms of progression-free survival (PFS) in patients with renal cell carcinoma who have progressed following 1-2 lines of therapy including an immune checkpoint inhibitor.
Dr. Figlin:
We all recognize that you’re to be congratulated along with Drs. Motzer, Dr. Albiges and other colleagues who are part of that study for addressing the critical unmet need. So let me just go around the table and ask each of you. We have accomplished so much in kidney cancer during the last two decades, through the era of the tyrosine kinase inhibitors and currently through the era of the immune checkpoint inhibitors and their combination strategies. I know that many of us are looking at triplets that we would never have thought about it in kidney cancer a couple of decades ago. Here we’re trying to combine it with agents such as HIF2a inhibitors for example. Where do you see us going from here Dr. Motzer? So how do you see the field evolving?
Dr. Motzer:
Theidentification of new agents with a novel mechanism of action is critical. One new class of drugs which Dr. Choueiri has been pivotal in terms of bringing forward are the HIF2a inhibitors. As a class these drugs are active and seem to be very well tolerated and combine well with other agents. In one s study with hypoxia-inducible factor 2α (HIF-2α) belzutifan plus cabozantinib, the preliminary results showed good antitumor activity and tolerability for the combination. Given the positive outcomes from tivozanib combination studies, as a next step I’d like to see one of the HIf2a inhibitors added to tivozanib and nivolumab as a first-line therapy. I’m even an advocate for a study of tivozanib plus nivolumab combination in the adjuvant setting. I recognize that TKIs as single agent certainly didn’t pan out in the adjuvant setting because of poor tolerability. However, tivozanib may have a better chance particularly in combination with a checkpoint inhibitor in the adjuvant setting. Lastly, we need a better understanding of underlying biology to see if we can identify patient subpopulation that will respond best to such combination settings.
Dr. Figlin:
Dr. Choueiri, without putting you on the spot, but putting you on the spot. You’ve articulated nicely an increasing role of IO-TKI as a second line after prior IO therapy in the high risk resected population. How do you think the field is going to evolve if there’s an increased uptake of immune checkpoint inhibition in the high risk resected population?
Dr. Choueiri:
Absolutely, this is why we need studies in the post IO setting. In TiNivo-2 which we are talking about, we may include a subgroup to assess outcomes in the prior adjuvant IO setting. I tend to believe it will not be different if patients progressed within a year after therapy. But it will be different biology if progression happens after a year. If this strategy is successful, then we have to look at that we have a drug approved with the same construct as the design of TiNivo-2, but strictly in the adjuvant setting. And that’s something we’ve been working on because what if the biology is different.
Dr. Figlin:
Absolutely. Dr. Albiges, one of the challenges is we still have to navigate through patients with brain metastases and bone metastases. Any insights on whether there are now evolving populations of patients that we need to be address because of unmet needs?
Dr. Albiges:
I agree with you that there is an unmet need in those patients with brain and/or bone metastasis and obviously, we need to think about those patients that are difficult to treat. Now, we know the role of multimodal approach combining stereotactic radiation therapy on top of our systemic treatment and maybe defining the optimal systemic treatment. it’s likely that we may want to combine VEGF-TKI with an immune checkpoint inhibitors so that we would be able to induce more tumor shrinkage in those subset of population. In addition to subset of patient with metastasis, there is also other tumor types such as non-clear cell renal cell carcinoma and other different tumor entities. For those, we clearly need to have more clinical trials being developed and test some of those combinations in such patient populations that usually have very aggressive features. So beyond clear cell RCC, there are challenges from different pathological, and specific tumors that need to be addressed. I really feel that we have made a long way but we still have a lot to go and especially with how to sequence those different agents and work on the rescue strategy.
Dr. Figlin:
Let me just summarize by saying that our distinguished colleagues have shared their insights regarding the survival benefits, tolerance profile, quality of life and the rationale for combining tivozanib with immunotherapy along with some key perspectives about prospective pivotal trials for both adjuvant as well as systemic therapeutic settings. I would like to thank Drs. Motzer, Choueiri and Albiges for joining with us for this stimulating roundtable discussion.
CONCLUDING REMARKS
An unmet need remains for developing novel therapeutic combination that produces effective, durable responses without adding substantial toxicity in patients with relapsed or refractory advanced RCC. Anti-angiogenic therapy in combination with ICIs in the first-line setting has demonstrated not only favorafble efficacy, but also improved tolerability in patients with advanced RCC. For example, tivozanib plus tivozanib combination demonstrated a promising safety and efficacy profile with minimal off-target effects as first-line and beyond treatment in patients with advanced RCC. In this roundtable discussion, renowned experts convened to examine the immunomodulatory potential of tivozanib and also its synergistic potential when combined with nivolumab as a treatment option in patients with treatment-naïve or previously treated metastatic RCC. Also, panelists shared their perspectives about the recent TIVO-3 and ongoing TiNivo-2 trial with regards to safety and efficacy.
CONFLICT OF INTERESTS
RJM reports consulting fees from Aveo, Calithera, Eisai, Eli Lilly, EMD Serono, Genentech, Merck, Novartis AG, Pfizer, and Roche, and contracted research to employer MSKCC for Bristol Myers Squibb, Eisai, Exelixis, Genentech, Merck, Pfizer, and Roche. TKC reports grants received from Pfizer during the conduct of the study; personal fees received from Agensys, Alexion, Alligent, American Society of Clinical Oncology, Analysis Group, AstraZeneca, Bayer, Bristol Myers Squibb, Celldex, Cerulean, Clinical Care Options, Corvus, Dana-Farber Cancer Institute, EMD Serono, Inc., Eisai, Exelixis, Foundation Medicine, Genentech/Roche, GSK, Harborside Press, Heron, Ipsen, Kidney Cancer Association, Kidney Cancer Journal, Lpath, Lancet Oncology, Lilly, Merck & Co., Michael J. Hennessy Associates, National Comprehensive Cancer Network, Navinata Health, New England Journal of Medicine, Novartis, Peloton Therapeutics, Pfizer, PlatformQ Health, Prometheus Laboratories, Sanofi, Seattle Genetics/Astellas, and UpToDate outside the conduct of the study; grants received from AstraZeneca, Bayer, Bristol Myers Squibb, Calithera, Cerulean, Corvus, Eisai, Exelixis, Foundation Medicine, Genentech/Roche, GSK, Ipsen, Merck & Co., Novartis, Peloton Therapeutics, Pfizer, Prometheus Laboratories, Takeda, and TRACON outside the conduct of the study; and medical writing and editorial assistance provided by ClinicalThinking, Envision Pharma Group, Fishawack Group of Companies, Health Interactions, and Parexel, funded by pharmaceutical companies. LA reports consulting fees compensated to their institution from Amgen, Astellas, AstraZeneca, Bristol Myers Squibb, Corvus Pharmaceuticals, Exelixis, Ipsen, Merck KGaA, Merck & Co., Novartis, Peloton Therapeutics, Roche, and Pfizer outside the submitted work. HM has declared no conflicts of interest. RAF: No relevant conflicts to report for this roundtable.
CONTRIBUTIONS
The roundtable panelists (authors) were invited to participate in this discussion by the journal. All authors listed in the manuscript contributed significantly to KCJ roundtable. All authors have read and approved the final version. The final content and article is the sole work of the authors.
ACKNOWLEDGMENTS
This educational roundtable program is supported byAVEO Oncology
For further information about TiNivo-2 study:TiNivo-2 clinical study
KEYWORDS:
Tivozanib, vascular endothelial growth factor receptor, tyrosine kinase inhibitor, nivolumab, immune checkpoint inhibitor, renal cell carcinoma, kidney cancer.
REFERENCES
- 1. FDA approves tivozanib for relapsed or refractory advanced renal cell carcinoma. Drug Approvals and Database 2021 (March 10, 2021). https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-tivozanib-relapsed-or-refractory-advanced-renal-cell-carcinoma.
- 2. Motzer RJ, Nosov D, Eisen T et al. Tivozanib versus sorafenib as initial targeted therapy for patients with metastatic renal cell carcinoma: results from a phase III trial. J Clin Oncol. 2013; 31: 3791-3799.
- 3. Rini BI, Pal SK, Escudier BJ et al. Tivozanib versus sorafenib in patients with advanced renal cell carcinoma (TIVO-3): a phase 3, multicentre, randomised, controlled, open-label study. Lancet Oncol. 2020; 21: 95-104.
- 4. Pal SK, Escudier B, Atkins MB, et al. TIVO-3: Final OS analysis of a phase III, randomized, controlled, multicenter, open-label study to compare tivozanib to sorafenib in subjects with metastatic renal cell carcinoma (RCC). Presented at: 2020 ASCO Virtual Program; May 27, 2020. Abstract 5062.
- 5. Pal SK, McDermott DF, Escudier et al. TIVO-3: Temporal characteristics of treatment-emergent adverse events and dose modifications with tivozanib and sorafenib in the phase 3 TIVO-3 study of relapsed or refractory mRCC. Presented at: 2021 ASCO Virtual Program; May 27, 2021. Abstract 4567.
- 6. Albiges L, Barthélémy P, M Gross-Goupil M, Negrier S, Needle MN, Escudier B. TiNivo: Safety and Effi cacy of Tivozanib-Nivolumab Combination Therapy in Patients With Metastatic Renal Cell Carcinoma. Ann. Oncol. 2021. 32(1), 97-102.
- 7. Voron T, Colussi O, Marcheteau E et al. VEGF-A modulates expression of inhibitory checkpoints on CD8+ T cells in tumors. J Exp Med. 2015; 212: 139-148.
- 8. Pawlowski N, et al. Impact of various first- and second-generation tyrosine-kinase inhibitors on frequency and functionality of immune cells. Cancer Res. 2013;73. Abstract 3971.
- 9. Winston W et al. Tivozanib, a selective VEGFR TKI, potently blocks angiogenesis and growth in tumors that express a high level of VEGF-C and are refractory to VEGF-A blockade. AACR–NCI–EORTC International Conference: Molecular Targets and Cancer Therapeutics; San Francisco, CA; November 12–16, 2011.
- 10. Choueiri TK, Albiges L, Hammers HJ, McKay RR, Heng DYC, Beckermann K, Kasturi V, and Motzer RJ. TiNivo-2: A phase 3, randomized, controlled, multicenter, open-label study to compare tivozanib in combination with nivolumab to tivozanib monotherapy in subjects with renal cell carcinoma who have progressed following one or two lines of therapy where one line has an immune checkpoint inhibitor. Journal of Clinical Oncology 2022 40:6_suppl, TPS405-TPS405.
- 11. Molina AM, Hutson TE, Nosov D, Tomczak P, Lipatov O, Sternberg CN, Motzer R, Eisen T. Efficacy of tivozanib treatment after sorafenib in patients with advanced renal cell carcinoma: crossover of a phase 3 study. Eur J Cancer. 2018 May;94:87-94. doi: 10.1016/j.ejca.2018.02.009. Epub 2018 Mar 20. PMID: 29547835; PMCID: PMC6774240.
- 12. Motzer RJ, Escudier B, McDermott DF, et al. Nivolumab versus Everolimus in Advanced Renal-Cell Carcinoma. N Engl J Med. 2015;373(19):1803-1813. doi:10.1056/NEJMoa1510665
- Correspondence to: Dr. Figlin,Robert.Figlin@cshs.org
The post Recent Advances in Tivozanib plus Nivolumab Combinatorial Strategies in Renal Cell Carcinoma first appeared on GUcancers.
]]>The post MEDICAL INTELLIGENCE – Kidney Cancer first appeared on GUcancers.
]]>Newsworthy, late-breaking kidney cancer information from Web-based sources, professional societies, and government agencies
FDA Approves Tivozanib as First Therapy for a Relapsed/Refractory Advanced RCC Subgroup
The first therapy for adults with relapsed or refractory advanced renal cell carcinoma who have received two or more prior systemic therapies has been granted approval by the FDA. This US FDA approval was granted based on the data from the phase 2 TIVO-3 clinical trial (NCT02627963). TIVO-3 is a controlled, multicenter, open-label, phase III trial of 350 patients with highly refractory metastatic RCC who had failed ≥2 prior regimens, including VEGF TKI treatment.
Lead investigator Dr. Brian Rini of this trial (NCT02627963) along with other senior investigator Dr. Thomas Hutson discussed the TIVO-3 outcomes and prospect of tivozanib for combinatorial therapy with other IO agents (See Page 4: Roundtable Discussion Section in this issue). Results that the hazard ratio for overall survival (OS) with tivozanib versus sorafenib was 0.97 (95% CI, 0.75-1.24; P =.78). The median OS in the tivozanib arm was 16.4 months (95% CI, 13.4-22.2) and 19.2 months in the sorafenib arm (95% CI, 15.0-24.2). The study included a subgroup of patients who received previous checkpoint inhibitor and VEGF inhibitor therapy, and in this population, the HR for death was 0.55 and was 0.57 for those who received 2 prior checkpoint or VEGF inhibitors. In terms of response, tivozanib led to an 18% (95% CI: 12%-24%) overall response rate compared with 8% (95% CI: 4%-13%) in the sorafenib arm. Tivozanib appeared to have a favorable safety profile during the study. Treatment-related adverse events (TRAEs) were observed in 84% compared with 94% of the sorafenib arm. Serious TRAEs were observed in 11% of the patients who received tivozanib compared with 10% of those treated with sorafenib.
Reference:
Rini BI, Pal SK, Escudier BJ, Atkins MB, Hutson TE, Porta C, Verzoni E, Needle MN, McDermott DF. Tivozanib versus sorafenib in patients with advanced renal cell carcinoma (TIVO-3): a phase 3, multicentre, randomised, controlled, open-label study. Lancet Oncol. 2020 Jan;21(1):95-104. PMID: 31810797.
FDA approves nivolumab/cabozantinib combo for frontline kidney cancer
On January 22, 2021, the Food and Drug Administration approved the combination of nivolumab (Opdivo, Bristol- Myers Squibb Co.) and cabozantinib (Cabometyx, Exelixis) as first-line treatment for patients with advanced renal cell carcinoma (RCC). The approval of nivolumab/cabozantinib combination regimen is based on findings from the phase 3 CheckMate-9ER trial (NCT03141177). Results indicated that the combination reduced the risk of disease progression or death by 49% versus sunitinib (Sutent) in treatment-naïve patients with advanced RCC, with a median progression-free survival of 16.6 months versus 8.3 months, respectively (HR, 0.51; P <.0001). The objective response rate (ORR) was also doubled with nivolumab/ cabozantinib in this setting compared with sunitinib, at 55.7% versus 27.1%, respectively (P <.0001). In the combination arm, the complete response (CR) rate was 8.0%, the partial response (PR) rate was 47.7%, and the stable disease (SD) rate was 32.2%. Additionally, 5.6% of patients had progressive disease (PD) and 6.5% were not evaluable or not assessed. In the sunitinib arm, the CR, PR, and SD rates were 4.6%, 22.6%, and 42.1%, respectively. Regarding safety, the incidence of the most common, any-grade and high-grade treatment-related adverse events (TRAEs) were similar in both arms. The overall rate of serious AEs was similar between the 2 groups; however, liver toxicity was more common with cabozantinib/nivolumab. Nineteen percent of patients on the combination required corticosteroids due to immune-related AEs, 4% of whom needed corticosteroids for at least 30 days.
Reference:
Choueiri TK, Powles T, Burotto M, et al. Nivolumab + cabozantinib vs sunitinib in first-line treatment for advanced renal cell carcinoma: first results from the randomized phase 3 CheckMate 9ER trial. Ann Oncol. 2020;31(4). Abstract 696O.
FDA Grants Belzutifan Priority Review for VHL-Associated RCC./
The novel, selective HIF-2 alpha inhibitor belzutifan was granted a priority review by the FDA for the treatment of patients with VHL–associated RCC who do not require immediate surgery. The primary end point of the study is ORR in VHL disease–associated RCC tumors and secondary end points include DOR, TTR, PFS, and time to surgery (TTS) in VHL disease– associated RCC tumors as well as ORR, DOR, TTR, PFS, and TTS in non-RCC tumors. This open-label phase 2 Study-004 trial (NCT03401788) supported the NDA, showing a significant response rate of 36.1% (95% CI, 24.2%-49.4%) in patients with VHL disease–associated RCC treated with belzutifan. Treatment-related adverse events (TRAEs) were observed in 96.7% of patients, most of which were grade 1 or 2 in severity; no grade 4 or 5 TRAEs were reported. The most common TRAEs were anemia in 83.6%, which was considered an on-target toxicity; fatigue in 49.2%; and dizziness in 21.3%. Grade 3 TRAEs, primarily fatigue and anemia, were reported in 9.8% of patients. Belzutifan is also being investigated in phase 3 trials as a monotherapy and in combination regimens in patients with RCC.
Reference:
Jonasch E, Donskov F, Iliopoulos O, et al. Phase II study of the oral HIF-2alpha inhibitor MK-6482 for Von Hippel-Lindau disease–associated renal cell carcinoma. J Clin Oncol. 2020;38(suppl 15):5003. doi:10.1200/ JCO.2020.38.15_suppl.5003.
Nivolumab Plus Ipilimumab Sparks Hope for Patients With RCC and Sarcomatoid Features.
Nivolumab plus ipilimumab combination therapy has improved survival and response rates compared with sunitinib, in patients with advanced renal cell carcinoma with sarcomatoid histology, including those with intermediate and poor-risk features. The post hoc, phase 3 CheckMate 214 clinical trial evaluated the efficacy of nivolumab (Opdivo) plus ipilimumab (Yervoy) versus sunitinib (Sutent) in patients with sRCC. 139 patients had sRCC and intermediate/poor-risk disease and 6 had favorable-risk disease from 1,096 included in the study. The study found that led to unprecedented long-term survival, response, and complete response when compared with sunitinib. Based on the results, investigators support the use of nivolumab plus ipilimumab as frontline treatment of patients with sRCC. PFS, on the other hand, was significantly longer with nivolumab plus ipilimumab at 26.5 months (95% CI, 8.4 to NE) compared with the 5.1 months (95% CI, 4.0-6.9) seen with sunitinib (HR, 0.54; 95% CI, 0.3-0.9; P = .0093). The median OS however was not reached with nivolumab plus ipilimumab (95% CI, 25.2 months–not estimable [NE]) versus 14.2 months (95% CI, 9.3-22.9) vs sunitinib (HR, 0.45; 95% CI, 0.3-0.7; P = .0004). Patients who received nivolumab with ipilimumab also achieved a higher ORR of 60.8% (95% CI, 49%-72%) compared with 23.1% (95% CI, 14%-35%) in the sunitinib arm (P < .0001). The complete response rate in the combination arm was 18.9% compared with only 3.1% in the control arm. “I believe patients with clear cell RCC, who have sarcomatoid features in the tumor should be, in my opinion, nivolumab and ipilimumab if you’re doing that for treatment in first line setting, I think the data we have from Checkmate-214 support this recommendation as the preferred first-line therapy for these patients” said Nizar Tannir, lead author of this trial.
Reference:
Tannir NM, Signoretti S, Choueri TK, et al. Efficacy and safety of nivolumab plus ipilimumab versus sunitinib in first-line treatment of patients with advanced sarcomatoid renal cell carcinoma. Clin Can Res. Published Online January 2021. Accessed February 3, 2021. https://bit.ly/36W2gSr.
Researchers unravel how kidney tumors’ microenvironments change in response to immunotherapy.
By using single-cell RNA sequencing, researchers from Dana-Farber Cancer Institute and the Broad Institute of MIT and Harvard investigated how kidney tumors’ microenvironments change in response to immunotherapy. Researchers discovered that in advanced stage disease these CD8+ T cells were “exhausted,” and not able to carry out their usual function. “These companion studies shed important new light on the biology of advanced kidney tumors and their surrounding environments. With this increased understanding, researchers will be able to identify new potential drug treatment targets and, overall, expand the number of patients who can receive effective treatment.” said Catherine J. Wu, MD, professor of medicine at Harvard Medical School. “A patient’s immune system plays a critical role in controlling both the progression of cancer and the response to immune therapies,” adds Toni K. Choueiri, MD, co-senior author of this paper. In other study, researchers performed single-cell RNA and T cell receptor sequencing on 164,722 individual cells from tumor and adjacent non-tumor tissue. They also discovered more anti- inflammatory or “M2-like” macrophages, a type of white blood cell that suppresses the immune system, in advanced stage disease. CD8+ T cells and macrophages were playing off each other and caught in an “immune dysfunction circuit,” said co-lead author David A. Braun, MD, PhD, an oncologist at Dana-Farber. “There may be immune evasion mechanisms outside of PD-1/PD-L1 that play an important role in response or resistance,” said Kevin Bi, computational biologist at Dana-Farber and co-lead author on the paper. Study found that immune dysfunction circuit is associated with a worse prognosis in external cohorts and identifies potentially targetable immune inhibitory pathways in ccRCC.
Reference:
Bi, K., et al. (2021) Tumor and immune reprogramming during immunotherapy in advanced renal cell carcinoma. Cancer Cell. doi.org/10.1016/j. ccell.2021.02.015.
The post MEDICAL INTELLIGENCE – Kidney Cancer first appeared on GUcancers.
]]>The post Guidelines for Kidney Cancer Treatment first appeared on GUcancers.
]]>
A list of global guidelines for kidney cancer are provided in this section. These guidelines provide evidence-based recommendations to serve as a guide and support best quality standards of care.
The post Guidelines for Kidney Cancer Treatment first appeared on GUcancers.
]]>