The post Kidney Cancer Research Highlights from ASCO 2022 Annual Meeting first appeared on GUcancers.
]]>
.
Updated
Yasser Ged,1 and Nirmish Singla2,*
1) Department of Oncology, The Johns Hopkins University School of Medicine, Baltimore, MD, USA
2) Department of Urology, The James Buchanan Brady Urological Institute, The Johns Hopkins University School of Medicine, Baltimore, MD, USA
ABSTRACT
The 2022 American Society of Clinical Oncology (ASCO) annual meeting was held June 3-7, 2022, in Chicago, Illinois. This hybrid meeting gathered international cancer experts across multidisciplinary specialties and was held both virtually and in-person. Here, we highlight key kidney cancer research updates presented at the meeting. Slides from the meeting’s presentations are available on the ASCO meeting library website.
INTRODUCTION
Adjuvant Therapy Updates Locally advanced kidney cancer has traditionally been managed surgically alone1. However, approximately 30% of patients develop recurrent metastatic disease after surgical resection despite curative intent, and the optimal approaches to integrate surgery with systemic therapies in a neoadjuvant or adjuvant approach to reduce the risk of recurrence has been an area of active research.2 The U.S. Food and Drug Administration (FDA) has approved two adjuvant therapies in renal cell carcinoma (RCC) thus far, including sunitinib in 2017 and most recently pembrolizumab in 2021.3,4 The use of adjuvant sunitinib has been limited despite FDA approval because of its increased toxicity and lack of overall survival benefit.5 Pembrolizumab is the first approved adjuvant immunotherapy for clear cell RCC patients with intermediate-high or high risk of recurrence after nephrectomy based on the phase 3 doubleblind, multicenter, randomized KEYNOTE-564 study (NCT03142334).4
Updated analysis from KEYNOTE-564 was presented at the meeting evaluating the time to first subsequent drug treatment or any-cause death (TFST) and time from randomization to progression on next line of therapy or any-cause death (PFS2) after treatment with pembrolizumab or placebo in the study.6 Overall 67 patients (13.5%) in the pembrolizumab group and 99 patients (19.9%) in the placebo group received ≥1 line of subsequent anticancer drug therapy. A total of 108 PFS2 events were observed, 40 (8.1%; 12 death events and 28 progression events) in the pembrolizumab group and 68 (13.7%; 14 death events and 54 progression events) in the placebo group. PFS2 was also delayed with pembrolizumab compared with placebo (HR, 0.57; 95% CI, 0.39-0.85; medians not reached). The authors concluded that treatment with adjuvant pembrolizumab reduced risk for TFST and PFS2 compared with placebo. LITESPARK-022 (NCT05239728) is the next iteration of the KEYNOTE-564 study which is a phase 3 study designed to compare the efficacy and safety of belzutifan plus pembrolizumab with that of placebo plus pembrolizumab as adjuvant treatment for clear cell RCC after nephrectomy, and this study is currently actively enrolling.

.
Multiple adjuvant and neoadjuvant vascular endothelial growth factor tyrosine kinase inhibitors (VEGF-TKIs) studies in RCC were reported previously.5 To better understand the role of mammalian target of rapamycin (mTOR) inhibitors in the adjuvant setting, the Southwest Oncology Group (SWOG) launched the phase 3 study of everolimus in treating patients with kidney cancer who have undergone surgery (EVEREST) study (NCT01120249), which was reported at ASCO 2022.7 Individuals with clear or non-clear cell RCC immediately post-nephrectomy whose tumors show intermediate high-risk to high risk features were included in the study. Between 4/2011 and 9/2016, 1545 patients were randomized to e ither 1 2 m onths o f a djuvant e verolimus (n = 7 75) o r placebo (n = 7 70) including 83% w ith clear cell RCC and 17% with non-clear cell RCC. With a median follow-up of 76 months, the recurrence free survival was improved with everolimus compared to placebo (HR 0.85, 95% CI, 0.72 – 1.00; P (one sided) = 0.0246), narrowly missing the pre-specified, one-sided significance level of 0.022 which accounted for interim analyses, and the effect of everolimus was especially pronounced in patients with very high risk disease. Adverse events were consistent with safety profiles of everolimus, although there was a high discontinuation rate of everolimus in this population (47%).

.
First Line Metastatic Kidney Cancer Treatment Updates The first line treatment landscape of metastatic RCC has rapidly evolved in recent years.8 New updates on some of the registration first line metastatic RCC studies were presented during the meeting. The CheckMate 9ER trial is a phase 3 trial which compared nivolumab plus cabozantinib versus sunitinib in patients with untreated advanced clear cell RCC and demonstrated superior overall survival (OS), progression free survival (PFS) and objective responses of the nivolumab plus cabozantinib combination9. Updated analysis from the depth of response was presented at ASCO 2022.10 Patients’ responses were classified as complete response (CR) or partial response (PR) subdivided by a tumor reduction of ≥80%–<100% (PR1), ≥60%–<80% (PR2), or ≥30%–<60% (PR3). Overall, greater proportions of patients receiving nivolumab plus cabozantinib had deeper responses versus sunitinib (CR, PR1, PR2), and deeper responses with nivolumab plus cabozantinib were associated with improved 12-months PFS rate versus sunitinib for CR (94.9% vs 82.4%), PR1 (81.3% vs 37.5%), and PR2 (72.1% vs 53.2%).
.
Updates on health-related quality of life (HRQoL) from the CheckMate-214 phase 3 clinical trial, which compared nivolumab plus ipilimumab versus sunitinib in patients with untreated advanced clear cell RCC, were also presented during the meeting.11,12 As previously reported, nivolumab plus ipilimumab was associated with improved HRQoL compared to sunitinib. At ASCO 2022, the investigators reported on a post-hoc analysis on the prognostic ability of HRQoL to inform the risk of disease progression or death. The results of the analysis showed that higher (better) baseline scores were associated with significantly reduced risk of death (HR [95% CI] for FKSI- 19 Total Score and DRS score was 0.83 [0.80-0.87] and 0.80 [0.76-0.84], respectively). Furthermore, patients with improved/stable HRQoL had a 52% reduction in risk of death compared to patients who had worsened (HR 0.48 [95% CI: 0.39-0.59]).
.
Post-hoc exploratory analyses of PFS2 were conducted in the KEYNOTE 426 (phase 3 study comparing pembrolizuamb plus axitinib versus sunitinib in patients with untreated advanced clear cell RCC)13,14 and the CLEAR (phase 3 study comparing pembrolizumab plus lenvatinib versus sunitinib in patients with untreated advanced clear cell RCC)15,16 studies. Both analyses demonstrated prolongation of PFS2 in patients who received pembrolizumab plus axitinib in KEYNOTE 426 study and pembrolizumab plus lenvatinib in the CLEAR study. Novel Kidney Cancer Therapies Highlights Several exciting data were presented on novel therapies in RCC. Batiraxcept is a GAS6-AXL inhibitor, a pathway which is overexpressed in clear cell RCC.17 Interim results of a phase 1b study of batiraxcept plus cabozantinib 60 mg daily were presented at the meeting.18 A total of 26 patients were enrolled in the phase 1b study so far, and the recommended phase 2 dose of batiraxcept was identified as 15 mg/kg every 2 weeks. Encouraging early anti-tumor efficacy results of the combination were observed with an objective response rate of 67% and 6 months PFS of 79%. Hypoxia-inducible factor 2α (HIF-2α) is a key oncogenic driver in RCC.19 Belzutifan is a HIF-2α inhibitor which was recently approved by the FDA for patients with VHL syndrome and currently under investigation in sporadic RCC.20,21 LITESPARK-001 is a phase 1 study which was designed to evaluate belzutifan in heavily pretreated RCC and showed durable antitumor activity and an acceptable safety profile.21 An update of the clear cell RCC cohort in the study with more than 3 years of total followup was presented at the meeting.22 With extended followup of 41 months, the objective response rate was 25% with 80% disease control rate and median PFS of 14.5 months (95% CI, 7.3-22.1). Belzutifan monotherapy continued to show a high rate of disease control and durable responses in this heavily pre-treated population.
The CALYPSO study results were presented at the meeting as well.23 This is a randomized phase II study of durvalumab alone or with savolitinib or tremelimumab in previously treated advanced clear cell RCC. Savolitinib is a potent MET inhibitor with established dosing and activity in papillary RCC; however, its role in clear cell RCC is unclear.24 Between 2017 and 2021, 139 patients were randomized across the treatment arms. Savolitinib alone and in combination with duravlumab was associated with modest confirmed response rates (5% and 13%, respectively) compared to confirmed response rates of 10% for durvalumab and 28% for durvalumab plus tremelimumab. All regimens studied in the trial appeared to be safe and tolerable.
SUMMARY
In summary, ASCO 2022 was enriched with novel results and concepts continually expanding the field of kidney cancer research. Indeed, the data presented are both hypothesis-generating and practice-informing. Herein, we highlighted a snapshot of some of the oral presentations from the meeting in the kidney cancer space; however, there are considerably more exciting abstract and poster presentations that are available for review on the meeting’s website. In addition to the scientific content, ASCO 2022 also provided ample opportunities for networking and collaborations among the academic kidney cancer community, with the first in-person option since the beginning of the COVID-19 pandemic.
.
REFERENCES
1. Campbell SC, Clark PE, Chang SS, Karam JA, Souter L, Uzzo RG. Renal Mass and Localized Renal Cancer: Evaluation, Management, and Follow-Up: AUA Guideline: Part I. J Urol. 2021;206(2):199-208.
2. Apolo AB, Msaouel P, Niglio S, et al. Evolving Role of Adjuvant Systemic Therapy for Kidney and Urothelial Cancers. Am Soc Clin Oncol Educ Book. 2022;42:1-16.
3. Mejean A, Ravaud A, Thezenas S, et al. Sunitinib Alone or after Nephrectomy in Metastatic Renal-Cell Carcinoma. N Engl J Med. 2018;379(5):417-427.
4. Choueiri TK, Tomczak P, Park SH, et al. Adjuvant Pembrolizumab after Nephrectomy in Renal-Cell Carcinoma. N Engl J Med. 2021;385(8):683-694.
5. Gleeson JP, Motzer RJ, Lee CH. The current role for adjuvant and neoadjuvant therapy in renal cell cancer. Curr Opin Urol. 2019;29(6):636-642.
6. Choueiri TK, Tomczak P, Park SH, et al. Adjuvant pembrolizumab for postnephrectomy renal cell carcinoma (RCC): Expanded efficacy analyses from KEYNOTE-564. 2022;40(16_suppl):4512-4512.
7. Ryan CW, Tangen C, Heath EI, et al. EVEREST: Everolimus for renal cancer ensuing surgical therapy—A phase III study (SWOG S0931, NCT01120249). 2022;40(17_suppl):LBA4500-LBA4500.
8. McKay RR, Bosse D, Choueiri TK. Evolving Systemic Treatment Landscape for Patients With Advanced Renal Cell Carcinoma. J Clin Oncol. 2018:JCO2018790253.
9. Choueiri TK, Powles T, Burotto M, et al. Nivolumab plus Cabozantinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med. 2021;384(9):829-841.
10. Suárez C, Choueiri TK, Burotto M, et al. Association between depth of response (DepOR) and clinical outcomes: Exploratory analysis in patients with previously untreated advanced renal cell carcinoma (aRCC) in CheckMate 9ER. 2022;40(16_suppl):4501-4501.
11. Motzer RJ, Tannir NM, McDermott DF, et al. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N Engl J Med. 2018;378(14):1277-1290.
12. Cella D, Hamilton M, Blum SI, et al. The relationship between health-related quality of life (HRQoL) and clinical outcomes in patients with advanced renal cell carcinoma (aRCC) in CheckMate (CM) 214. 2022;40(16_suppl):4502-4502.
13. Rini BI, Plimack ER, Stus V, et al. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med. 2019;380(12):1116-1127.
14. Powles T, Plimack ER, Stus V, et al. Pembrolizumab (pembro) plus axitinib (axi) versus sunitinib as first-line therapy for advanced clear cell renal cell carcinoma (ccRCC): Analysis of progression after first subsequent therapy in KEYNOTE-426. 2022;40(16_suppl):4513-4513.
15. Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus Pembrolizumab or Everolimus for Advanced Renal Cell Carcinoma. N Engl J Med. 2021;384(14):1289-1300.
16. Voss MH, Powles T, McGregor BA, et al. Impact of subsequent therapies in patients (pts) with advanced renal cell carcinoma (aRCC) receiving lenvatinib plus pembrolizumab (LEN + PEMBRO) or sunitinib (SUN) in the CLEAR study. 2022;40(16_suppl):4514-4514.
17. Rankin EB, Fuh KC, Castellini L, et al. Direct regulation of GAS6/AXL signaling by HIF promotes renal metastasis through SRC and MET. Proc Natl Acad Sci U S A. 2014;111(37):13373-13378.
18. Shah NJ, Beckermann K, Vogelzang NJ, et al. A phase 1b/2 study of batiraxcept (AVB-S6-500) in combination with cabozantinib in patients with advanced or metastatic clear cell renal cell (ccRCC) carcinoma who have received front-line treatment (NCT04300140). 2022;40(16_suppl):4511-4511.
19. Choueiri TK, Kaelin WG, Jr. Targeting the HIF2-VEGF axis in renal cell carcinoma. Nat Med. 2020;26(10):1519-1530.20. Jonasch E, Donskov F, Iliopoulos O, et al. Belzutifan for Renal Cell Carcinoma in von Hippel- Lindau Disease. N Engl J Med. 2021;385(22):2036-2046.
21. Choueiri TK, Bauer TM, Papadopoulos KP, et al. Inhibition of hypoxia-inducible factor-2alpha in renal cell carcinoma with belzutifan: a phase 1 trial and biomarker analysis. Nat Med. 2021;27(5):802-805.
22. Jonasch E, Bauer TM, Papadopoulos KP, et al. Phase 1 LITESPARK-001 (MK-6482-001) study of belzutifan in advanced solid tumors: Update of the clear cell renal cell carcinoma (ccRCC) cohort with more than 3 years of total follow-up. 2022;40(16_suppl):4509-4509.
23. Powles T, Mendez-Vidal MJ, Rodriguez-Vida A, et al. CALYPSO: A three-arm randomized phase II study of durvalumab alone or with savolitinib or tremelimumab in previously treated advanced clear cell renal cancer. 2022;40(17_suppl):LBA4503-LBA4503.
24. Choueiri TK, Heng DYC, Lee JL, et al. Efficacy of Savolitinib vs Sunitinib in Patients With METDriven Papillary Renal Cell Carcinoma: The SAVOIR Phase 3 Randomized Clinical Trial. JAMA Oncol. 2020;6(8):1247-1255.
Correspondence to:
Nirmish Singla, MD, MSCS. Departments of Urology and Oncology, The James Buchanan Brady Urological Institute, The Johns Hopkins School of Medicine. Baltimore MD 21287.
EMAIL: nsingla2@jhmi.edu
The post Kidney Cancer Research Highlights from ASCO 2022 Annual Meeting first appeared on GUcancers.
]]>The post ASCO21 Meeting Summary – Kidney Cancer first appeared on GUcancers.
]]>
Ochsner Precision Cancer Therapies Program and Ochsner Cancer Institute, Benson Cancer Center. New Orleans LA
Correspondence to: Marc R. Matrana. Ochsner Precision Cancer Therapies Program and Ochsner Cancer Institute 1514 Jefferson Highway, New Orleans LA – 70121. E-mail: MaMatrana@ochsner
ABSTRACT
For the second year in a row, the annual meeting of the American Society of Clinical Oncology (ASCO) was held virtually due to the ongoing COVID-19 pandemic. Nonetheless, the meeting was hailed as a great success and brought much practice changing data in the field of genitourinary medical oncology, including kidney cancer.
Figure: ASCO21 Virtual Meeting participants. Courtesy: Luca Dezzani
Adjuvant Therapy in RCC
The most ground-breaking presentation was given by Dr. Toni Choueiri, who presented interim results of Keynote-564, a phase 3 double-blind trial comparing adjuvant pembrolizumab to placebo following nephrectomy in clear cell RCC patients with intermediateand high-risk of recurrence. The study enrolled 994 patients with pT2 grade 4 or sarcomatoid, pT3-4 any grade, node positive disease (any grade, any T stage) RCC within 12 weeks of nephrectomy and those with completely resected oligometastatic disease within a year of nephrectomy. The great majority of patients fell into the first two categories. The results represented the first prespecified analysis after approximately 265 disease free survival (DFS) events. DFS at 12 months was 76.2 in the control arm vs 85.7% in pembrolizumab arm and at 24 months was 68.1 vs 77.3% in those arms. This represented a hazard ratio (HR) of 0.68 (95% CI: 0.53-0.87, p=0.0010). Overall survival (OS) was not mature and only included about a quarter of events, but still trended towards a meaningful improvement with a 46% reduction in the risk of death in those receiving pembrolizumab, HR= 0.54 (95% CI 0.30 – 0.96, p=0.0164). Safety was as expected with no new signals emerging. This represents the first positive adjuvant immunotherapy trial in RCC and in the opinion of many in the genitourinary medical oncology community, the first step towards a viable adjuvant option in RCC, given the overall weak efficacy and unfavorable toxicity profile of sunitinib in this setting. Yet, there is still more work to be done. As noted above, the OS was not mature and many questions remain, including role of PD-L1 status as a biomarker of adjuvant benefit of immunotherapy, how non-clear cell might respond to this approach, and the benefit patients with early stage disease or who are more than 12 weeks from nephrotomy might benefit, among others. Fortunately, several ongoing studies are investing some of these issues and the Keynote-564 data continues to mature.
Novel Therapies and Combinations Yuan-Yuan Qu and colleagues from China provided impressive updated results from a phase II study of the anti- PD-L1 camrelizumab plus famitinib, a TKI against VEGFR-2, PDGFR, c-kit, and FGFR, in patients with advanced, heavily pre-treated RCC and urothelial carcinoma. Thirty-eight patients with advanced RCC were included, with an ORR of 63.2% (95% CI, 46.0- 78.2; 24 PRs), DCR of 89.5% (95% CI, 75.9-95.8), and median DOR which was not reached (range 2-19+ mos) in this cohort. Median PFS was also not reached, and 12-mo OS rate was 88.0%. 92.1% of subjects had reduction in target lesions with a median reduction of 47% from baseline noted. DCR was 100% (95% CI, 77.2-100.0) in untreated RCC patients and 84.0% (95% CI, 65.3- 93.6) in pre-treated patients. Further study of this regimen certainly seems warranted.
Results of a phase 1b study of the novel IL-2v immunocytokine Simlukafusp α were presented by Perez-Garcia et al. This study enrolled 69 patients with unresectable clear cell and/or sarcomatoid RCC who were treated with the novel drug paired with atezolizumab or bevacizumab or both. The triplet combination was found to be most efficacious with ORR of 47- 48% including 3 complete responses in patients treated with triplet therapy.
Dr. Nizar Tannir of MD Anderson presented results of the highly anticipated CANTATA study, which randomized 444 patients who had progressed on 1-2 prior lines of therapy to a combination of the glutaminase inhibitor telaglenastat plus cabozantinib or placebo plus cabozantinib. Unfortunately, the study did not meet its endpoints. Median PFS (mPFS) was 9.2 months for telaglenastat plus cabozantinib arm vs 9.3 months for the control arm (HR = 0.94; 95% CI: 0.74, 1.21; stratified log-rank p= 0.65) with overall response rates (ORR; confirmed) of 31% with Tela+Cabo vs 28% Pbo+Cabo, respectively.
Tumor growth in Von Hippel- Lindau disease is driven by an aberrant accumulation of HIF-2alpha due to the inactivation of VHL, ultimately resulting in the development of clear cell RCC, pancreatic neuroendocrine tumors (pNET), and hemangioblastomas. A phase 2 study of the HIF-2alpha inhibitor belzutifan (MK-6482) in patients with Von Hippel-Lindau disease associated RCC revealed promising results. 61 patients were enrolled with a median follow-up of 69 weeks, of those, 92% remained on therapy at the time of analysis. There were 22 confirmed responses (36%) and (11%) unconfirmed responses. ORR was 80% in pNETs including one complete response and 32% in CNS hemangioblastomas, also including one complete response. There were 16 retinal hemangioblastoma patients evaluable at baseline, of which 69% showed improvement.
There is much research around the role of the microbiome in cancer, and particularly around response to immunotherapy. Results of a randomized phase 1b study of CBM- 588 (an agent principally consisting of Clostridium butyricum) showed a significantly improved ORR when this drug was added nivolumab plus ipilimumab (59%) as compared to nivolumab plus ipilimumab alone (11%). Median PFS was also prolonged (NR vs 11 weeks), although sample size was small (30 subjects) and the comparator arm did much poorer than historically expected.
Xinan Sheng and colleagues from China presented a Phase 3 double-blind three arm trial in which randomized patients received in a 1:1:1 fashion either the VEGF-targeted vorolanib plus placebo, everolimus, or a combination of these. 399 patients were enrolled. Median PFS was prolonged in combination arm compared to the single-agent everolimus group (10.0 months [95% CI, 8.2-10.4] vs. 6.4 months [95% CI, 4.7-8.3]; HR = 0.70 [95% CI, 0.52-0.94]; P = 0.0171). Median PFS was similar between single-agent vorolanib and single-agent everolimus (6.4 months [95% CI, 4.6-8.3] vs. 6.4 months [95% CI, 4.7-8.3]; HR = 0.94 [95% CI, 0.69- 1.24]; P = 0.6856). Response was achieved by 33/133 (24.8%) of patients in the combination arm, 11/133 (8.3%) in the single-agent everolimus arm, and 14/133 (10.5%) of those receiving singleagent vorolanib. OS was immature at the time of the analysis. The combination was associated with Grade 3 or greater treated related AEs in 72% of subjects.
Trial Updates
Final results of the phase 3 KEYNOTE-426 study of pembrolizimab plus axitinib vs sunitinib in metastatic RCC the first line setting were presented. Previous analysis showed the combination significantly improved OS, PFS, and ORR vs sunitinib monotherapy in treatment-naive advanced clear cell RCC and the combination gained FDA approval. Overall, 861 subjects were randomized with 418 deaths at this 42-month analysis. The combination of pembrolizumab plus axitinib improved OS compared to sunitinib (median: 45.7 vs 40.1 mo; HR, 0.73 [95% CI: 0.60- 0.88]; P<0.001) and PFS (median: 15.7 vs 11.1 mo; HR, 0.68 [95% CI, 0.58- 0.80]; P<0.0001). The 42-mo OS rate was 57.5% with combination vs 48.5% in the control arm; the 42-mo PFS rate was 25.1% with the combination vs 10.6% with sunitinib. ORR for the combination was 60.4% vs 39.6% with suntinib (P<0.0001), and a CR rate of 10.0% vs 3.5% in the combination arm and control arm, respectively. No new safety signals were observed in this long-term analysis.
Investigators p r e s e n t e d updated results focusing on the efficacy of nivolumab + i p i l imu m a b in IMDC intermediate and poor risk a d v a n c e d clear cell RCC patients with initial versus late progression with nivolumab from the TITAN-RCC study. In this tailored immunotherapy approach, patients who did not respond initially were given between 2-4 doses of nivolumab + ipilimumab as boost cycles and patients who had PR or CR initially on nivolumab monotherapy were eligible for nivolumab + ipilimumab at progression. Primary endpoint was objective response rate in first line and second line whereas the secondary endpoints included activity in nivolumab monotherapy, remission rate of patients who receive the nivolumab + ipilimumab boost and safety and overall survival. Results showed that the TITAN tailored immunotherapy approach improved response rates in patients compared to those patients who had received only nivolumab monotherapy with nearly half of patients receiving nivolumab + ipilimumab boosts with PD improving to PR/CR (18%) or SD (30%). Health-related quality-of-life analysis from the phase 3 CLEAR study was presented by Robert Motzer. The trial, which showed that lenvatinib plus pembrolizumab improved PFS, OS, and ORR compared to sunitinib, also found the combination of lenvatinib plus pembrolizumab was associated with similar better symptoms and quality of life than sunitinib. Improvements in physical functioning, fatigue, dyspena, and constipation were significantly improved with the combination when compared with sunitinib.
Updated data on the durability of response and overall survival from the TIVO-3 study of tivozanib versus sorafenib were provided. The study enrolled patients with metastatic RCC who failed 2 or 3 prior systemic therapies, one of which included a VEGFR-targeted TKI. There were 41 responders (23%) in the tivozanib and 20 responders (11%) in the sorafenib. The median duration of response was 20.3 months (95% CI: 9.8 – 29.9) with tivozanib and and 9.0 months (95% CI: 3.7 -16.6) with sorafenib. The HR for overall survival favored tivozanib at 0.91 (95% CI: 0.716 – 1.165).
An interesting study of outcomes of second-line therapy patients who progressed on the JAVELIN Renal 101 study was presented by Laurence Albiges. She and her colleagues analyzed outcomes of 163 patients who received single agent therapy (including 60 who received cabozantinib) and 41 patients who received combination therapy (most commonly levantinib and everolimus) following avelumab plus axitinib on study. 36 months OS was 44.1% in those receiving single-agent therapy (95% CI: 35.9 – 52.0) and 63.4 months (95% CI: 45.7 – 76.6) in those receiving combination therapy in the second-line setting. Median secondline PFS was 20.4 months (95% CI: 17.6 – 23.0) and 24.1 months (95% CI: 17.7 – NE) in these two groups, respectively.
Comparisons of First Line Therapies
For many physicians, the question of which first-line combination therapy to use in patients with newly diagnosed metastatic RCC is daunting given the number of new combinations approved. This is exacerbated by a lack of head-tohead comparison studies among these combinations. Two ASCO abstracts attempted to compare regimens outside of the scope of a prospective head-tohead trial.
Kevin Zarrabi and colleagues abstracted data from 821 mRCC patients from Flatiron databases, 259 who received axitinib plus pembrolizumab and 562 who received ipilimumab plus nivolumab. Demographics and clinical parameters were well matched between the two cohorts. Median age was 66 years, 73% were male, and 54.9% had a nephrectomy. The adjusted median OS between the two treatment groups was n ot s tatistically d ifferent. T welvemonth survival was 68.5% for those receiving axitinib plus pembrolizumab and 65.8% for received ipilimumab plus nivolumab treated patients (P=0.41). Twelve-month real world PFS was 41.4% for the axitinib plus pembrolizumab group and 39.7% for those in the received ipilimumab plus nivolumab group (P=0.14).
Bradley Alexander McGregor and colleagues take a slightly different approach using a matching-adjusted indirect comparison (MDIC) of data from the Checkmate 9ER and Keynote-426 studies of nivolumab plus cabozantinib and pembrolizumab plus axitinib respectively. Nivolumab plus cabozantinib was associated with a median PFS of 19.3 compared to 15.7 months with pembrolizumab plus axitinib, and nivolumab plus cabozantinib also achieved superior overall response rate (ORR) and duration of response (DoR). OS outcomes were similar. Cytoreducitve Nephrectomy The role of cytoredutive nephrectomy in RCC in the targeted and immunotherapy era continues to be debated. An abstract by Jaleh Fallah and colleagues explored nephrectomy prior to immunotherapy based combinations via a pooled analysis of data from 5 trials. Among 849 patients with Stage IV RCC at initial diagnosis, 62% had nephrectomy prior to immunotherapy based combinations. Median OS was not reached in the prior nephrectomy group, but appeared prolonged in those having surgery prior to systemic immunotherapy, even when adjusted for age and prognostic risk scores (HR=0.59, 95% CI: 0.46-0.75). In a similar retrospective study, Pooja Ghatalia et al extracted data on 1,719 mRCC patients from the Flatiron Health database, and found that 972 (56.5%) received systemic treatment alone, 605 (35.1%) received cytoreductive nephrectomy followed by systemic treatment, and 142 (8.2%) received systemic followed by cytoreductive nephrectomy. Cytoreductive nephrectomy prior to systemic therapy was significantly associated with prolonged OS compared to systemic therapy alone, HR=0.82, (95% CI: 0.70-0.95, p=0.008). Taken together, these two retrospective studies suggest that cytoreductive nephrectomy may play a role in improving outcomes in certain patients and that still more studies of this controversial area are needed, especially as the landscape of system therapy continues to evolve.
Non-Clear Cell RCC
Several abstracts reported data on therapies for non-clear cell RCC. Due to the rarity of metastatic collecting duct carcinoma, the BONSAI trial was opened as a phase 2 evaluating the primary endpoint of objective response rate of patients taking cabozantinib 60mg orally once daily. From January 2018 to November 2020, 25 patients were enrolled and 23 were treated. Six patients had stable disease, 1 with a CR, and 7 with a PR. Secondary endpoints showed an ORR of 35% and median PFS of 6 months. Treatment showed optimistic efficacy and acceptable patient tolerability in this patient population.
Gopalakrishnan et al. conducted a retrospective analysis of 203 patients with renal cell carcinoma with any sarcomatoid component and had received systemic therapy from 6 US academic cancer centers evaluating the overall survival and response rates. It was found that median overall survival and response rates were significantly higher in patients who had previously received immune checkpoint inhibitors compared to those patients who had not. For instance, median overall survival was 31% with immune checkpoint inhibitor versus 7.6% and a 66.2% disease control rate with immunotherapy versus 39.1% nonimmunotherapy drugs. Additionally, there was a large benefit noted among patients who had non clear cell and mixed histology sarcomatoid renal cell carcinoma.
In the Oracle study, investigators wanted insight to determine if there was antitumor activity with new combination therapies such as IO, VEGF, and mTOR inhibitors in patients with metastatic nCCRCC. Research shows that despite this patient population having inferior rates with new novel combination therapies than clear cell RCC, there is some antitumor activity observed which warrants further prospective studies.
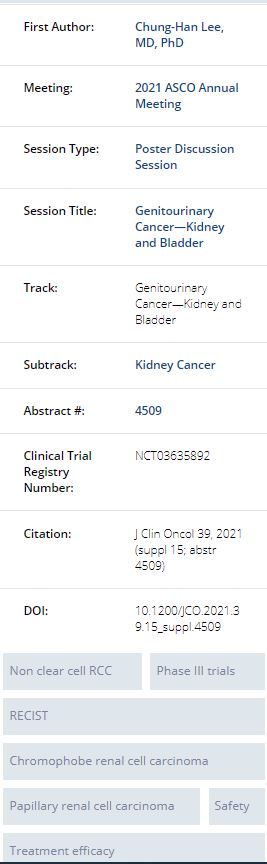
Rodriguez et al. evaluated the combination of savolitinib 1500mg every 4 weeks and durvalumab 600mg every day in MET-driven, metastatic papillary renal cell carcinoma in a single arm phase I/II trial. Investigators analyzed the confirmed response rate, PFS, tolerability and overall survival of the 41 patients who received treatment. The confirmed response rate in MET driven patients was 57% with a duration of response of 9.4 months, and a median PFS of 10.5 months. OS was 27.4 months (95% CI: 7.3-NR). Clinical activity of this combination seems promising for MET driven papillary renal cell carcinoma patients. The results of a phase 2 trial of patients with advanced nccRCC on Cabozantinib and nivolumab were presented by Lee et al. Patients with no history of prior immune checkpoint inhibitors and up to 1 prior line of therapy with measurable disease received cabozanitinib 40mg plus nivolumab 240mg every 2 week versus 480mg every 4 weeks in two different cohorts. Cohort 1 consisted of 40 patients with papillary, unclassified, or translocation associated RCC and cohort 2 consisted of 7 patients with chromophobe histology. Overall results showed that there is promising efficacy in metastatic non-clear cell RCC pts with papillary, unclassified, or translocation associated histologies. ORR for the papillary, unclassified, or translocation cohort was 48% (95% CI 31.5–63.9; Table), and a median PFS of 12.5 months (95% CI 6.3–16.4) and median OS was 28 months (95% CI 16.3–NE). Unfortunately, none of the 7 chromophobe patients has a response suggesting there is limited efficacy in this sub-population.
In part 2 of UNISON (ANZUP 1602), investigators evaluated the results of treating patients with rare variant RCC refractory to single-agent nivolumab with salvage ipilimumab and nivolumab. 41 patients were determined to be refractory to single agent nivolumab and had variant of nccRCC histologies. One complete response and 3 partial responses were noted. Disease control rate at 6 months was 45% with a median PFS of 2.6 months. The primary endpoint of this study was not met. The results from this study show that although a small portion of nccRCC refractory to nivolumab may benefit from combination therapy with ipilimumab and nivolumab, the majority do not, and more research is needed to identify valuable treatment options for nccRCC patients.
Potential Biomarkers
The search for biomarkers in RCC continues. Soleimani et al evaluated plasma exosome microRNAs (miRNAs) as a potential biomarker of response to immune checkpoint-based therapy. Prior to initiating immunotherapy, 11 miRNAs that are over-expressed in RCC and/or immune-associated were evaluated in 40 patients and in 30 healthy volunteers. RT-PCT was used to evaluate MiRNA expression between the two groups and compared using the 2DDCt method. The most common first line immunotherapy was nivolumab + ipilimumab, followed by pembrolizumab + axitinib, and avelumab + axitinib. Results showed that a lower expression of miR-155 was associated with response to immunotherapy in patients with metastatic RCC and highlight the need for additional research in this area for use of miR-155 as a biomarker of response. Previously, the 27-gene immuno-oncology signature has been shown to be associated with value to immune checkpoint inhibitors in breast, lung, and bladder cancers. Investigators aimed to identify if this could also be applied to patients with renal cell carcinoma. Patients who had a positive score by the 27-gene signature had significantly better one-year PFS compared to patients with a negative score (hazard ratio = 0.235, 95% CI = 0.069 – 0.803, p < 0.01). Given that four tumor types validated this algorithm, additional research is needed to support this assay as a pan-cancer immune system classifier.
Tucker et al presented a retrospective review of patients seeing to evaluate and correlate baseline neutrophil-to-eosinophil ratio (NER) and neutrophil-to-lymphocyte ratio (NLR) with treatment outcomes with ipilimumab and nivolumab in patients with metastatic RCC. 111 patients were included in the analysis and it was found that patient with NER less than median had prolonged median PFS, OS, ORR as compared to those with NER greater than the median. Data suggested that baseline NER may serve as an early response predictor for immunotherapy with ipilimumab and nivolumab.
Sites of Metastases
Previous studies have described RCC with metastatic disease to the pancreas as a distinct clinical entity with a more indolent course as compared to historical controls. Cassandra Duarte and colleagues presented an analysis of 229 patients from 9 institutions. They report a median OS for all patients from time of metastatic disease was prolonged at 7.7 years (95% CI 6.3-10.3). The median OS for those who received a first line VEGF targeted therapy was 7.6 years (95% CI 5.5-9.5) and was the median OS was not reached for those receiving first-line immunotherapy (95%CI 6.5-NR). The difference was significant with an unadjusted p-value of 0.029, with a pair-wise comparison between median OS of first line immunotherapy subgroup compared to that of the first line VEGF inhibitor group also achieving statistical significance (p = 0.0148). These results suggest the choice of first-line therapy in mRCC with pancreatic metastases may impact outcome.
Patients with brain metastases are often excluded from clinical trials, limiting data on outcomes in this populations. CheckMate 920 is an ongoing phase 3b/4 trial of nivolumab and ipilimumab in patients with advanced RCC with unmet medical need, including those with asymptomatic brain metastases. Of the 25 mRCC patients with brain metastases who were evaluable for response, the ORR was 32.0% (95% CI, 14.9–53.5). The median duration of response was 24.0 months with 50% of responders without reported progression.
Oligoprogressive Disease
Raquibul Hannan et al presented data from a prospective phase II single arm trial of mRCC patients who demonstrated response to systemic therapy and then had subsequent radiographic evidence of three or fewer sites of disease progression were treated. These patients underwent stereotatic ablative radiation (SAbR) to all progressive sites. The study found that SAbR extended PFS by > 6 months in 70.6% of the 17 evaluable oligoprogressive mRCC patients. SAbR was safe and did not adversely affect QOL, suggesting it is a reasonable alterative to switching systemic therapies in patients with limited progression in a small number of metastatic sites.
Healthcare Disparities
Janvi Wadiwala and colleagues highlight access issues with regards to palliative care among RCC patients. In a National Cancer Database study of 50,405 patients found that socioeconomic barriers such as age, race, Spanish/Hispanic origin, income, education, and other factors affected metastatic renal cell carcinoma patients access to palliative care resources. Older patients, African Americans, higher income, and lower education status patients were less likely to receive palliative care. High education attainment and treatment at an integrated cancer center or comprehensive community cancer center increased the odds of receiving palliative care. Gender differences in health outcomes represent another important area of healthcare disparity research, Claud Grigg and colleagues explored survival trends between men and women with metastatic RCC. Clear cell renal cell carcinoma is twice as common in men as in women, and previous studies have shown improved outcomes in women with localized RCC as compared to men. In this retrospective NCDB study, patients were grouped by date of metastatic diagnosis into three eras, pre-Tyrosine Kinase Inhibitor (TKI), TKi and immune checkpoint inhibitors (ICI). Demographic features were similar, with females being slightly older and more likely to be black. Women were found to be at an increased risk of death in both the ICI era and the TKI era, with a worse prognosis than men that could not be explained by demographic differences.
Patient Voice
Results from a global survey of 2,012 patients and caregivers from 41 countries were reported by Rachel H. Giles and colleagues. They found more than half (52%) of the responders lacked understanding of subtype of diagnosis, but also reported that a similar number (51%) felt they were as involved as they wanted to be with their care. Between 41-45% reported the no one discussed clinical trials with them and the no one explained their likelihood of surviving their cancer beyond 5 years. These results help guide provider attention to areas where patient comprehension and needs can be better addressed. COVID-19 in RCC
Of course, last but certainly not least is the impact that the COVID-19 pandemic has had on RCC patients. A retrospective study by Jesus Garcia Donas et al compared patients with advanced RCC who developed COVID while undergoing antiangiogenic treatment vs immunotherapy vs matched controls. Patients with RCC who developed COVID19 had treatment held more frequently and presented with lower clinical benefit rates than those not infected. Those patients on immunotherapy required more frequent interruptions and longer hospitalizations than those on antiangiogenics.
As the world continues to address the global pandemic, cancer has not slowed down. Thankfully, the fight against cancer also hasn’t slowed down either as evidenced by the remarkable progress exemplified in the ASCO 2021 Annual Meeting. It is our hope that we can all join together once again in Chicago for ASCO 2022.



The post ASCO21 Meeting Summary – Kidney Cancer first appeared on GUcancers.
]]>The post ASCO21 Sessions: RECOMMENDED ABSTRACTS in Renal Cancer first appeared on GUcancers.
]]>
These recommended abstracts have been selected by Robert A. Figlin, MD, Editor-in- Chief of the Kidney Cancer Journal. The chosen abstracts provided here highlight some of the most important trends in ongoing trials and reflect the foremost research and strategies from latest clinical trials that impact the current standard of care in renal cancer.

ABSTRACT 4500:
ASCO Link – https://meetinglibrary.asco.org/record/195569/abstract
Pembrolizumab (pembro) plus axitinib (axi) versus sunitinib as first-line therapy for advanced clear cell renal cell carcinoma (ccRCC): Results from 42-month follow-up of KEYNOTE-426
Brian I. Rini, Elizabeth R. Plimack, Viktor Stus, Tom Waddell, Rustem Gafanov, Frédéric Pouliot, Dmitry Nosov, Bohuslav Melichar, Denis Soulieres, Delphine Borchiellini, Ihor O. Vynnychenko, Raymond S. McDermott, Sergio Jobim Azevedo, Satoshi Tamada, Anna Kryzhanivska, Chenxiang Li, Joseph E. Burgents, L. Rhoda Molife, Jens Bedke, Thomas Powles; Vanderbilt-Ingram Cancer Center, Nashville, TN; Fox Chase Cancer Center, Philadelphia, PA; Dnipropetrovsk Medical Academy of Ministry of Health of Ukraine, Dnipro, Ukraine; The Christie NHS Foundation Trust, Manchester, United Kingdom; Russian Scientific Center of Roentgenoradiology, Moscow, Russian Federation; CHU of Québec and Laval University, Québec City, ON, Canada; Central Clinical Hospital With Outpatient Clinic, Moscow, Russian Federation; Palacky University Medical School and Teaching Hospital, Olomouc, Czech Republic; Centre Hospitalier de l’Université de Montréal, Montréal, QC, Canada; Centre Antoine Lacassagne, Université Côte d’Azur, Nice, France; Sumy State University, Sumy Regional Oncology Center, Sumy, Ukraine; Adelaide and Meath Hospital and University College Dublin, Dublin, Ireland; Hospital de Clínicas de Porto Alegre, Porto Alegre, Brazil; Osaka City University Hospital, Osaka, Japan; Ivano-Frankivsk National Medical University, Ivano-Frankivsk, Ukraine; Merck & Co., Inc., Kenilworth, NJ; MSD UK, London, United Kingdom; Eberhard Karls Universität Tübingen, Tübingen, Germany; Barts Health and the Royal Free NHS Trusts, Barts Cancer Institute, Queen Mary University of London, London, United Kingdom
Background: In the first interim analysis of the randomized, multicenter, open-label, phase 3 KEYNOTE-426 study (NCT02853331), treatment with pembro + axi significantly improved OS, PFS, and ORR vs sunitinib monotherapy in treatment-naive advanced ccRCC. Extended follow-up (median, 30.6 mo) continued to demonstrate the superior efficacy of pembro + axi vs sunitinib monotherapy in this patient population. Here, we present the results of the prespecified final analysis with 42.8-mo median follow-up.
Methods: Treatment-naive patients (pts) with advanced ccRCC, KPS ≥70%, and measurable disease (RECIST v1.1) were randomly assigned 1:1 to receive pembro 200 mg IV Q3W for up to 35 doses + axi 5 mg orally BID or sunitinib 50 mg orally QD on a 4-wk on/2-wk off schedule until progression, intolerable toxicity, or withdrawal. Randomization was stratified by IMDC risk (favorable vs intermediate vs poor) and geographic region (North America vs Western Europe vs Rest of World). Dual primary endpoints were OS and PFS. Secondary endpoints were ORR, DOR, and safety. The protocol-specified final analysis was based on a target of 404 OS events. No formal hypothesis testing was performed because all efficacy endpoints were met previously at the first interim analysis; nominal P values are reported.
Results: Overall, 861 pts were randomly assigned to receive pembro + axi (n=432) or sunitinib (n=429). Median duration of follow-up, defined as time from randomization to the database cutoff date, was 42.8 mo (range, 35.6-50.6). At data cutoff, 418 pts had died: 193 (44.7%) of 432 pts in the pembro + axi arm vs 225 (52.4%) of 429 pts in the sunitinib arm. Compared with sunitinib, pembro + axi improved OS (median: 45.7 vs 40.1 mo; HR, 0.73 [95% CI, 0.60-0.88]; P<0.001) and PFS (median: 15.7 vs 11.1 mo; HR, 0.68 [95% CI, 0.58-0.80]; P<0.0001). The 42-mo OS rate was 57.5% with pembro + axi vs 48.5% with sunitinib; the 42-mo PFS rate was 25.1% with pembro + axi vs 10.6% with sunitinib. For pembro + axi vs sunitinib, ORR was 60.4% vs 39.6% (P<0.0001); CR rate was 10.0% vs 3.5%; median DOR was 23.6 mo (range 1.4+ to 43.4+) vs 15.3 mo (range, 2.3-42.8+). Subsequent anticancer therapy was administered to 47.2% of pts in pembro + axi arm vs 65.5% of pts in sunitinib arm. Although a similar proportion of pts in each arm received VEGF/VEGFR inhibitors, only 10.2% of pts in the pembro + axi arm received subsequent treatment with a PD-1/L1 inhibitor compared to 48.7% of pts in the sunitinib arm. No new safety signals were observed. Conclusions: With a median follow-up of 42.8 mo, this is the longest follow-up of an anti-PD–1/L1 immunotherapy combined with a VEGF/VEGFR inhibitor for first-line RCC. These results show that pembro + axi continues to demonstrate superior efficacy over sunitinib with respect to OS, PFS, and ORR, with no new safety signals.
Clinical trial information: NCT02853331
Research Funding: Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA 

ABSTRACT 4501 :
ASCO Link – https://meetinglibrary.asco.org/record/195568/abstract
CANTATA: Primary analysis of a global, randomized, placebo (Pbo)-controlled, double-blind trial of telaglenastat (CB-839) + cabozantinib versus Pbo + cabozantinib in advanced/metastatic renal cell carcinoma (mRCC) patients (pts) who progressed on immune checkpoint inhibitor (ICI) or anti-angiogenic therapies.
Nizar M. Tannir, Neeraj Agarwal, Camillo Porta, Nicola Jane Lawrence, Robert J. Motzer, Richard J. Lee, Rohit K. Jain, Nancy B. Davis, Leonard Joseph Appleman, Oscar B. Goodman, Walter Michael Stadler, Sunil G. Gandhi, Daniel M. Geynisman, Roberto Iacovelli, Begona Mellado, Robert A. Figlin, Thomas Powles, Lalith V Akella, Keith W. Orford, Bernard Escudier; The University of Texas MD Anderson Cancer Center, Houston, TX; Huntsman Cancer Institute, University of Utah, Salt Lake City, UT; University of Bari ‘A. Moro’ and Policlinico Consorziale di Bari, Bari, Italy; Auckland City Hospital, Auckland, New Zealand; Memorial Sloan Kettering Cancer Center, New York, NY; Massachusetts General Hospital, Boston, MA; H. Lee Moffitt Cancer Center & Research Institute, Tampa, FL; Vanderbilt-Ingram Cancer Center, Nashville, TN; University of Pittsburgh Medical Center, Pittsburgh, PA; Comprehensive Cancer Centers of Nevada, Las Vegas, NV; The University of Chicago, Chicago, IL; Florida Cancer Specialists, Lecanto, FL; Fox Chase Cancer Center, Department of Hematology and Oncology, Philadelphia, PA; Fondazione Policlinico Universitario A. Gemelli IRCCS, Rome, Italy; Hospital Clínic, Provincial de Barcelona, Barcelona, Spain; Cedars-Sinai Medical Center, Samuel Oschin Comprehensive Cancer Institute, Los Angeles, CA; St Bartholomew’s Hospital, Barts Health NHS Trust, London, United Kingdom; Calithera Biosciences, Inc., South San Francisco, CA; Gustave Roussy, Villejuif, France
Background:Dysregulated metabolism is a hallmark of RCC, driven by overexpression of glutaminase (GLS), a key enzyme of glutamine metabolism. Telaglenastat (Tela) is an investigational, first-in-class, selective, oral GLS inhibitor that blocks glutamine utilization and critical downstream pathways. Preclinically, Tela synergized w/ cabozantinib (Cabo), a VEGFR2/MET/AXL inhibitor, against RCC tumors. In a Ph 1 study cohort, Tela+Cabo showed encouraging safety/efficacy as 2L+ therapy for mRCC. This trial compared Tela+Cabo vs Pbo+Cabo in previously treated pts w/ clear-cell mRCC (NCT03428217).
Methods: Eligible pts had 1-2 prior lines of systemic therapy for mRCC, including ≥1 anti-angiogenic therapy or nivolumab + ipilimumab (nivo/ipi), KPS ≥70%, measurable disease (RECIST 1.1), no prior Cabo or other MET inhibitor. Pts were randomized 1:1 to receive Cabo (60 mg PO QD) with either Tela (800 mg PO BID) or Pbo, until disease progression/unacceptable toxicity, and were stratified by prior PD-(L)1 inhibitor therapy (Y/N) and IMDC prognostic risk group. Primary endpoint was progression-free survival (PFS; RECIST 1.1) by blinded independent radiology review. The study was designed to detect a PFS hazard ratio (HR) of 0.69 w/ alpha 0.05 and 85% power. Data cutoff date: August 31, 2020.
Results: 444 pts were randomized (221 Tela+Cabo; 223 Pbo+Cabo). Baseline characteristics were balanced between arms. Median follow-up was 11.7 mo; 276 pts received prior ICI, including 128 w/ prior nivo/ipi. Median PFS (mPFS) was 9.2 mo for Tela+Cabo vs 9.3 mo for Pbo+Cabo (HR = 0.94; 95% CI: 0.74, 1.21; stratified log-rank P= 0.65) with overall response rates (ORR; confirmed) of 31% with Tela+Cabo vs 28% Pbo+Cabo, respectively. Overall survival was not mature at data cutoff. In a prespecified subgroup analysis in pts w/ prior ICI, mPFS was numerically longer w/ Tela+Cabo than Pbo+Cabo (11.1 vs 9.2 mo, respectively; unstratified HR = 0.77; 95% CI: 0.56, 1.06). In the Pbo+Cabo arm, mPFS was 9.2 mo for pts w/ prior ICI exposure and 9.5 mo for pts without, and ORR was 32% and 20%, respectively; if ICI included nivo/ipi, ORR was 37%. Rates of adverse events (AEs) were similar between arms.Grade 3-4 AEs occurred in 71% of Tela+Cabo pts and 79% of Pbo+Cabo pts and included hypertension (17% vs 18%) and diarrhea (15% vs 13%). Cabo was discontinued due to AEs in 10% of Tela+Cabo pts and 15% of Pbo+Cabo pts.
Conclusions: The addition of Tela did not improve the efficacy of Cabo in mRCC in this study. Tela+Cabo was well tolerated with AEs consistent with known risks of both agents. The study provides valuable insight on efficacy outcomes of a contemporary population of pts w/ mRCC who receive Cabo in the 2/3L setting.
Clinical trial information: NCT03428217
Research Funding: Calithera Biosciences, Inc


LATE BREAKTHROUGH ABSTRACT LBA 5 :
ASCO Link – https://meetinglibrary.asco.org/record/196683/abstract
Pembrolizumab versus placebo as post-nephrectomy adjuvant therapy for patients with renal cell carcinoma: Randomized, double-blind, phase III KEYNOTE-564 study.
Toni K. Choueiri, Piotr Tomczak, Se Hoon Park, Balaji Venugopal, Tom Ferguson, Yen-Hwa Chang, Jaroslav Hajek, Stefan N. Symeonides, Jae-Lyun Lee, Naveed Sarwar, Antoine Thiery-Vuillemin, Marine Gross-Goupil, Mauricio Mahave, Naomi B. Haas, Piotr Sawrycki, Eric (Pingye) Zhang, Jaqueline Willemann Rogerio, Kentaro Imai, David I. Quinn, Thomas Powles; Dana-Farber Cancer Institute, Boston, MA; Szpital Kliniczny Przemienienia Pańskiego UM, Poznan, Poland; Division of Hematology-Oncology, Samsung Medical Center, Department of Medicine, Seoul, South Korea; University of Glasgow, Glasgow, United Kingdom; Royal Perth Hospital, Perth, Australia; Department of Urology, Taipei Veterans General Hospital, Taipei, Taiwan; Fakultni Nemocnice Ostrava, Ostrava, Czech Republic; Edinburgh Cancer Research Centre, University of Edinburgh, Edinburgh, United Kingdom; Asan Medical Center and University of Ulsan College of Medicine, Seoul, South Korea; Imperial College Healthcare NHS Trust, London, United Kingdom; University Hospital Jean Minjoz, Besançon, France; Centre Hospitalier Universitaire de Bordeaux-Hôpital Saint-André, Bordeaux, France; Lopez Perez Foundation, Santiago, Chile; Abramson Cancer Center, University of Pennsylvania (ECOG-ACRIN), Philadelphia, PA; Wojewodzki Szpital Zespolony im. L. Rydygiera w Toruniu, Torun, Poland; Merck & Co., Inc., Kenilworth, NJ; USC Norris Cancer Hospital, Los Angeles, CA; Barts Cancer Institute, Cancer Research UK Experimental Cancer Medicine Centre, Queen Mary University of London, Royal Free National Health Service Trust,, London, United Kingdom
Background:Relapse after surgery for high-risk clear cell RCC (ccRCC) is associated with shortened life expectancy. Effective perioperative therapy to reduce this risk remains an unmet need. Adjuvant immune therapy is an attractive potential strategy for these pts. We conducted the KEYNOTE-564 trial to evaluate pembro vs placebo as adjuvant therapy for pts with RCC.
Methods: KEYNOTE-564 is a phase III multicenter trial of pembro vs placebo in pts with histologically confirmed ccRCC, with intermediate-high risk (pT2, Gr 4 or sarcomatoid, N0 M0; or pT3, any Gr, N0 M0), high risk (pT4, any Gr, N0 M0; or pT any stage, any Gr, N+ M0), or M1 NED (no evidence of disease after primary tumor + soft tissue metastases completely resected ≤1 year from nephrectomy) (Leibovich et al, 2003; Fuhrman et al, 1982). Pts had undergone surgery ≤12 wks prior to randomization; had no prior systemic therapy; had ECOG PS 0 or 1. Study treatment was given for up to 17 cycles (≈1 yr). The primary endpoint was disease-free survival (DFS) per investigator assessment in all randomized pts (ITT population). Overall survival (OS) was a key secondary endpoint. Safety/tolerability were secondary endpoints, assessed in all treated pts.
Results: Between Jun 30, 2017 and Sept 20, 2019, 994 pts were randomized 1:1 to pembro (n=496) or placebo (n=498). As of data cutoff date of Dec 14, 2020, median (range) follow-up, defined as time from randomization to data cutoff, was 24.1 (14.9−41.5) mo. No pts remain on study treatment. Baseline characteristics were generally balanced between arms. At first prespecified interim analysis, the primary endpoint of DFS was met (median not reached [NR] for both arms, HR 0.68, 95% CI 0.53−0.87; P=0.0010 [one-sided]). The estimated DFS rate at 24 mo was 77.3% with pembro vs 68.1% with placebo. Overall, DFS benefit was consistent across subgroups. A total of 51 OS events were observed (18 in the pembro arm, 33 in the placebo arm). Median OS was NR for both arms (HR 0.54, 95% CI 0.30−0.96; P=0.0164 [one-sided]); the p-value did not cross the statistical hypothesis testing boundary. The estimated OS rate at 24 mo was 96.6% with pembro vs 93.5% with placebo. 470 pts (96.3%) and 452 pts (91.1%) experienced ≥1 all-cause adverse events (AEs) with pembro vs placebo, respectively. Grade 3-5 all-cause AEs occurred in 158 pts (32.4%) with pembro and 88 pts (17.7%) with placebo. No deaths related to pembro occurred.
Conclusions: Pembro demonstrated a statistically significant and clinically meaningful improvement in DFS vs placebo in pts with intermediate-high, high risk or M1 NED RCC. Additional follow-up is planned for the key secondary endpoint of OS. KEYNOTE-564 is the first positive phase III study with a checkpoint inhibitor in adjuvant RCC, and these results support pembro as a potential new standard of care for pts with RCC in the adjuvant setting.
Clinical trial information: NCT03142334
Research Funding: Eisai Inc., Woodcliff Lake, NJ, USA, and Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA




ABSTRACT 4502 :
ASCO Link -https://meetinglibrary.asco.org/record/196538/abstract

Health-related quality-of-life (HRQoL) analysis from the phase 3 CLEAR trial of lenvatinib (LEN) plus pembrolizumab (PEMBRO) or everolimus (EVE) versus sunitinib (SUN) for patients (pts) with advanced renal cell carcinoma (aRCC).
Robert J. Motzer, Camillo Porta, Boris Alekseev, Sun Young Rha, Toni K. Choueiri, Maria Jose Mendez-Vidal, Sung-Hoo Hong, Anil Kapoor, Jeffrey C. Goh, Masatoshi Eto, Jinyi Wang, Janice Pan, Alemseged Ayele Asfaw, Cixin Steven He, Kalgi Mody, David Cella; Memorial Sloan Kettering Cancer Center, New York, NY; San Matteo University Hospital Foundation, Pavia, Italy; P.A. Herzen Moscow Oncological Research Institute, Moscow, Russian Federation; Yonsei Cancer Center, Yonsei University Health System, Seoul, South Korea; Dana-Farber Cancer Institute, Boston, MA; Maimonides Institute for Biomedical Research of Cordoba (IMIBIC), Hospital Universitario Reina Sofía, Córdoba, Spain; Seoul St. Mary’s Hospital, The Catholic University of Korea, Seoul, South Korea; McMaster University Hamilton, Hamilton, ON, Canada; ICON Research, South Brisbane & University of Queensland, St Lucia, QLD, Australia; Kyushu University, Fukuoka, Japan; RTI Health Solutions, Research Triangle Park, NC; Eisai Inc., Woodcliff Lake, NJ; Merck & Co., Inc., Kenilworth, NJ; Northwestern University, Chicago, IL
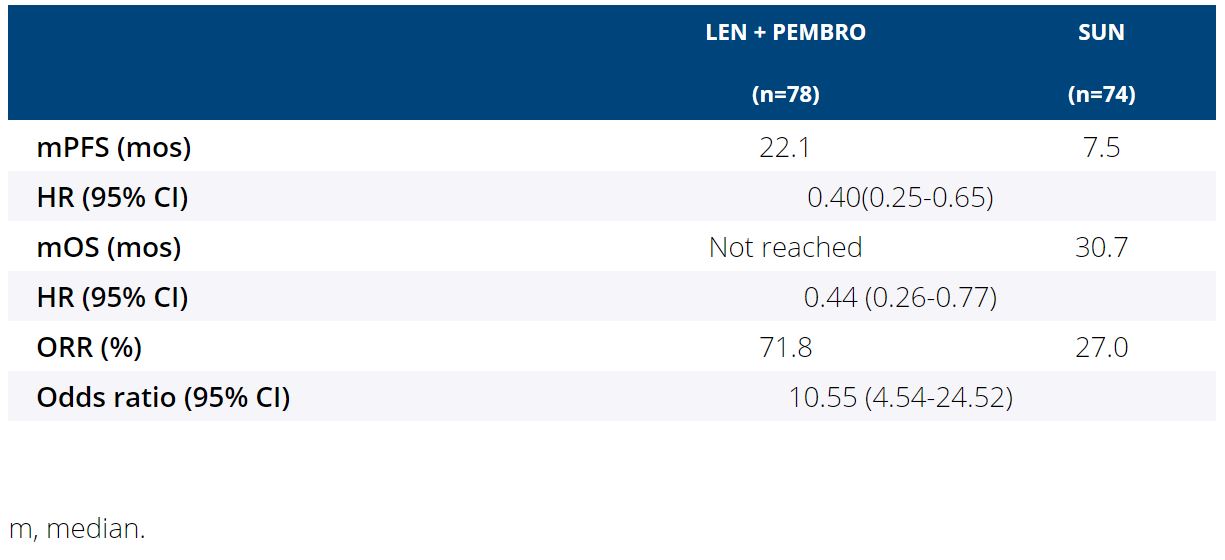
Background:LEN + PEMBRO improved PFS, OS and ORR vs SUN in the first-line treatment of pts with aRCC; LEN + EVE improved PFS and ORR vs SUN (Motzer R et al. NEJM. 2021). We report results of a secondary objective of the CLEAR trial comparing the impact of LEN + PEMBRO or EVE vs SUN, on HRQoL.
Methods: Pts (N=1069) were randomized (1:1:1) to receive LEN 20 mg PO QD + PEMBRO 200 mg IV Q3W; LEN 18 mg + EVE 5 mg PO QD; or SUN 50 mg PO QD (4 wks on/2 wks off). HRQoL was assessed per FKSI-DRS, EORTC QLQ-C30, and EuroQoL EQ-5D-3L, at baseline, on day 1 of subsequent 3 wk cycles starting with cycle 2, and at the off-treatment visit. HRQoL analyses (unless otherwise noted) were based on data from randomized pts with any HRQoL data who received ≥1 dose of study treatment. No adjustments for multiple testing or estimation were used; P-values and CIs are nominal and descriptive.
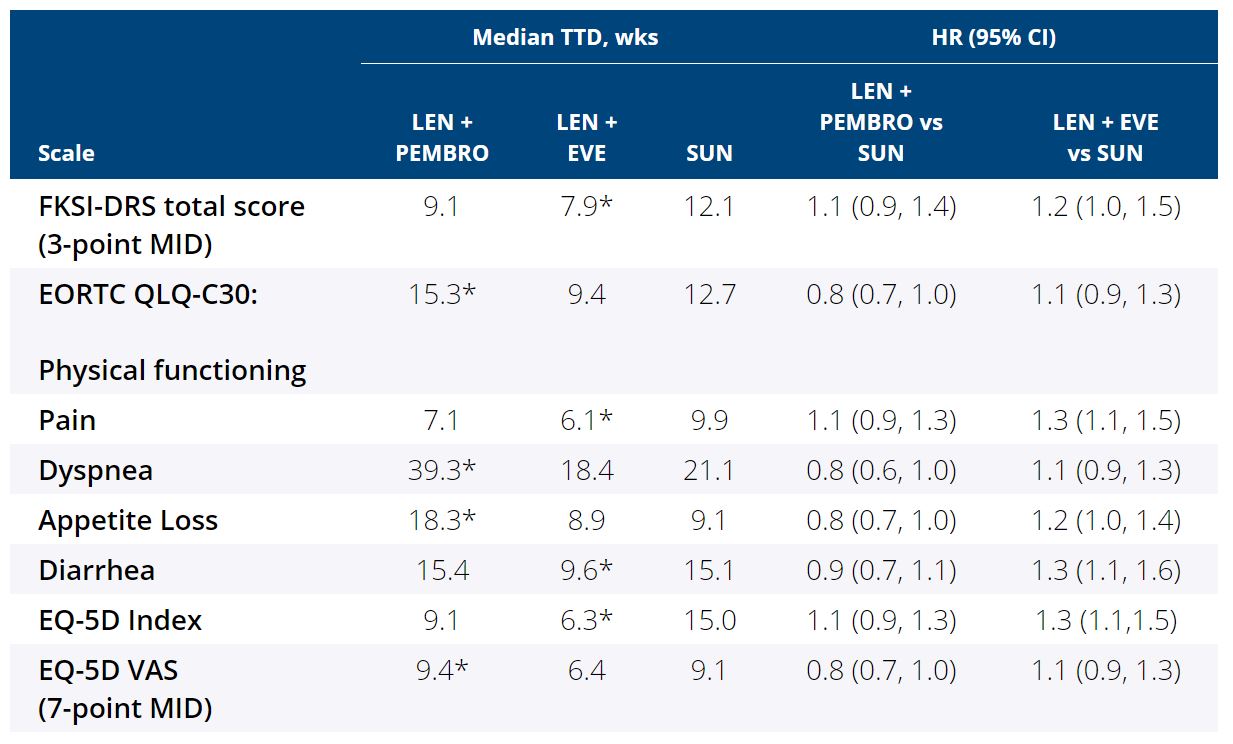
Results: For comparisons of LEN + PEMBRO vs SUN, overall changes from baseline at mean follow-up (wk 46) favored LEN + PEMBRO with significant differences between treatments for physical functioning (least squares mean difference [LS MD] [95% CI]: 3.0 [0.5, 5.5]) and fatigue (−2.8 [−5.5, −0.1]), dyspnea (−2.8 [−5.3, −0.3]), and constipation (−2.2 [−4.2, −0.2]). LS MD of the FKSI-DRS total score was 0.2 (−0.4, 0.7). For comparisons of LEN + EVE vs SUN, overall changes from baseline at wk 46 favored SUN with significant differences in overall HRQoL (−2.8 [−5.1, −0.5] assessed by the EORTC QLQ-C30 GHS/QoL scale) and pain (2.8 [0.1, 5.5]), appetite loss (4.2 [1.3, 7.1]), and diarrhea (5.3 [2.6, 7.9]). LS MD of the FKSI-DRS total score was −0.4 (−1.0, 0.2). 14 of 18 scales for both LEN + PEMBRO and LEN + EVE vs SUN had no significant differences in LS MD comparisons. The LEN + PEMBRO arm is favored over SUN for the median time to first deterioration (TTD) for physical functioning, dyspnea, appetite loss and EQ-5D VAS (Table). 15 of 19 scales for both LEN + PEMBRO and LEN + EVE vs SUN had no significant differences in TTD comparisons.
Conclusions: Compared with SUN, pts in LEN + PEMBRO group had similar or better symptoms and HRQoL.
Clinical trial information: NCT02811861
Research Funding: Eisai Inc., Woodcliff Lake, NJ, USA, and Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA


ABSTRACT 4509 :
ASCO Link – https://meetinglibrary.asco.org/record/195580/abstract
Nivolumab plus cabozantinib in patients with non-clear cell renal cell carcinoma: Results of a phase 2 trial.
Chung-Han Lee, Martin H Voss, Maria Isabel Carlo, Ying-Bei Chen, Eduard Reznik, Andrea Knezevic, Robert A Lefkowitz, Natalie Shapnik, Diana Tassone, Chloe Dadoun, Neil J. Shah, Colette Ngozi Owens, Deaglan Joseph McHugh, David Henry Aggen, Andrew Leonard Laccetti, Ritesh Kotecha, Darren R. Feldman, Robert J. Motzer; Memorial Sloan Kettering Cancer Center, New York, NY; Columbia University Medical Center, New York, NY; MD Anderson Cancer Center, Houston, TX
Background:Cabozantinib plus nivolumab (CaboNivo) improved objective response rate (ORR), progression-free survival (PFS), and overall survival (OS) over sunitinib in a phase 3 trial for metastatic clear cell renal cell carcinoma (RCC). (Choueiri, abstract 6960, ESMO 2020) We report the results of a phase 2 trial of CaboNivo in patients (pts) with non-clear cell RCC.
Methods: Pts had advanced non-clear cell RCC, 0 or 1 prior systemic therapies excluding prior immune checkpoint inhibitors, and measurable disease by RECIST. Cabo 40 mg/day plus Nivo 240 mg every 2 weeks or 480 mg every 4 weeks was given across two cohorts. Cohort 1: papillary, unclassified, or translocation associated RCC; Cohort 2: chromophobe RCC. The primary endpoint was ORR by RECIST; secondary endpoints included PFS, OS, and safety. Cohort 1 was a single stage design that met its primary endpoint and was expanded to produce more precise estimates of ORR. Cohort 2 was a Simon two-stage design that closed early for lack of efficacy. Correlative analyses by next generation sequencing were performed and to be presented.
Results: A total of 40 pts were treated in Cohort 1, and 7 pts were treated in Cohort 2 (data cutoff: Jan 20, 2021). Median follow up time was 13.1 months (range 2.2 – 28.6). In Cohort 1, 26 (65%) pts were previously untreated, and 14 (35%) pts had 1 prior line: 10 (25%) received prior VEGF-targeted therapy and 8 (20%) received prior mTOR-targeted therapy. ORR for Cohort 1 was 48% (95% CI 31.5–63.9; Table). Median PFS was 12.5 months (95% CI 6.3–16.4) and median OS was 28 months (95% CI 16.3–NE). No responses were seen among 7 patients in Cohort 2 with chromophobe histology (Table). Grade 3/4 treatment emergent adverse events were consistent with that reported in the phase 3 trial; Grade 3/4 AST and ALT were 9% and 15%, respectively. Cabozantinib and nivolumab were discontinued due to toxicity in 17% and 19% of pts, respectively.
Conclusions: CaboNivo had an acceptable safety profile and showed promising efficacy in metastatic non-clear cell RCC pts with papillary, unclassified, or translocation associated histologies whereas activity in patients with chromophobe RCC was limited.
Clinical trial information: NCT03635892
Research Funding: Exelixis, BMS


ABSTRACT 4555 :
ASCO Link – https://meetinglibrary.asco.org/record/197609/abstract
Phase 2 study of belzutifan (MK-6482), an oral hypoxia-inducible factor 2α (HIF-2α) inhibitor, for Von Hippel-Lindau (VHL) disease-associated clear cell renal cell carcinoma (ccRCC).
Ramaprasad Srinivasan, Frede Donskov, Othon Iliopoulos, Wendy Kimryn Rathmell, Vivek Narayan, Benjamin L. Maughan, Stephane Oudard, Tobias Else, Jodi K. Maranchie, Sarah Joanne Welsh, Ananya Roy, Yanfang Liu, Rodolfo F. Perini, W. Marston Linehan, Eric Jonasch; Center for Cancer Research, National Cancer Institute, Bethesda, MD; Aarhus University Hospital, Aarhus, Denmark; Massachusetts General Hospital Cancer Center and Harvard Medical School, Boston, MA; Vanderbilt-Ingram Cancer Center, Nashville, TN; University of Pennsylvania, Philadelphia, PA; University of Utah, Salt Lake City, UT; Hôpital Européen Georges Pompidou, Paris, France; University of Michigan, Ann Arbor, MI; University of Pittsburgh, Pittsburgh, PA; Cambridge University Hospitals NHS Foundation Trust, Cambridge, United Kingdom; Merck & Co., Inc., Kenilworth, NJ; The University of Texas MD Anderson Cancer Center, Houston, TX
Background:Inactivation of VHL leads to aberrant stabilization and accumulation of HIF-2α, which drives tumor growth. Patients (pts) with VHL disease are at risk for ccRCC, pancreatic neuroendocrine tumors (pNETs), and hemangioblastomas. Repeated surgeries are often needed to control ccRCC and other VHL disease manifestations. Prior results of this ongoing open-label phase 2 study (NCT03401788) showed activity with belzutifan in VHL disease. Updated results are presented.
Methods: Adults with germline VHL alterations, measurable and localized/nonmetastatic ccRCC, no prior systemic anticancer therapy, and ECOG PS 0 or 1 received belzutifan 120 mg once daily until progression, intolerable toxicity, or decision to withdraw. The primary end point is ORR of VHL-associated ccRCC tumors per RECIST v1.1 by independent review committee (IRC). Secondary end points include DOR, time to response (TTR), PFS, and safety
Results: As of June 1, 2020, 61 pts enrolled. Most pts (82%) had ECOG PS 0, and the median number of prior tumor reduction procedures (eg, partial nephrectomy, craniotomy, radiation therapy) per pt was 5 (range, 0-15). Lesions outside the kidney (non-RCC tumors) evaluable by IRC included pNETs (33%) and CNS hemangioblastomas (82%). Median follow-up was 69 wk (range, 18-105), median duration of treatment was 68 wk (range, 8-105), and 56 pts (92%) remain on therapy. There were 22 confirmed responses (ORR, 36% [95% CI, 24-49]) and 7 (11%) unconfirmed responses (documented at 1 time point, to be confirmed at subsequent time point); all were PRs. In pts with confirmed PR, median DOR was not reached (range, 12+ to 62+ wk), median TTR was 31 wk (range, 12-61), and 56 pts (92%) had some reduction in the sum of all target lesion diameters. PFS rate at 52 wk was 98% (95% CI, 89-100). For non-RCC tumors, ORR was 80% (16/20; 1 CR) in pNETs and 32% (16/50; 1 CR) in CNS hemangioblastomas. Of 16 pts with evaluable retinal hemangioblastomas at baseline, 11 (69%) showed improvement per IRC. In those 16 pts, 29 eyes were monitored for retinal hemangioblastomas: 16 eyes (55%) showed improvement, 12 (41%) were stable, and no evaluation was available for 1 eye (3%). All 61 pts (100%) had at least one AE. The most common all-cause AE was anemia (90%), which is considered an on-target toxicity. Treatment-related AEs (TRAE) were reported by 60 pts (98%), and 8 pts (13%) had a grade 3 TRAE. No pts had grade 4/5 TRAEs. One pt discontinued treatment because of a TRAE (grade 1 dizziness).
Conclusions: Belzutifan demonstrates clinical benefit and has a favorable safety profile in patients with VHL disease–associated ccRCC, pNETs, and hemangioblastomas.
Clinical trial information: NCT03401788
Research Funding: Research Funding: Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA 

ABSTRACT 4562:
ASCO Link – https://meetinglibrary.asco.org/record/197543/abstract
Post hoc analysis of the CLEAR study in advanced renal cell carcinoma (RCC): Effect of subsequent therapy on survival outcomes in the lenvatinib (LEN) + everolimus (EVE) versus sunitinib (SUN) treatment arms.
Thomas E. Hutson, Toni K. Choueiri, Robert J. Motzer, Sun Young Rha, Anna Alyasova, Jaime R. Merchan, Howard Gurney, Avivit Peer, Toshio Takagi, Camillo Porta, Thomas Powles, Viktor Grünwald, Ugo De Giorgi, Ulka N. Vaishampayan, Manuela Schmidinger, Hilary Glen, Karla Rodriguez-Lopez, Dongyuan Xing, Lea Dutta, Masatoshi Eto; Texas Oncology, Dallas, TX; Dana-Farber Cancer Institute, Boston, MA; Memorial Sloan Kettering Cancer Center, New York, NY; Yonsei Cancer Center, Yonsei University Health System, Seoul, South Korea; Prevoljskiy Region Medical Centre, Novgorod, Russian Federation; University of Miami Sylvester Comprehensive Cancer Center, Miami, FL; Macquarie University Hospital, Sydney, NSW, Australia; Rambam Health Care Campus, Haifa, Israel; Tokyo Women’s Medical University, Tokyo, Japan; San Matteo University Hospital Foundation, Pavia, Italy; The Royal Free NHS Trust, London, United Kingdom; University Hospital Essen, Essen, Germany; Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori (IRST) IRCCS, Meldola, Italy; University of Michigan, Ann Arbor, MI; Medical University of Vienna, Vienna, Austria; Beatson West of Scotland Cancer Center, Glasgow, United Kingdom; Merck & Co., Inc., Kenilworth, NJ; Eisai Inc., Woodcliff Lake, NJ; Kyushu University, Fukuoka, Japan
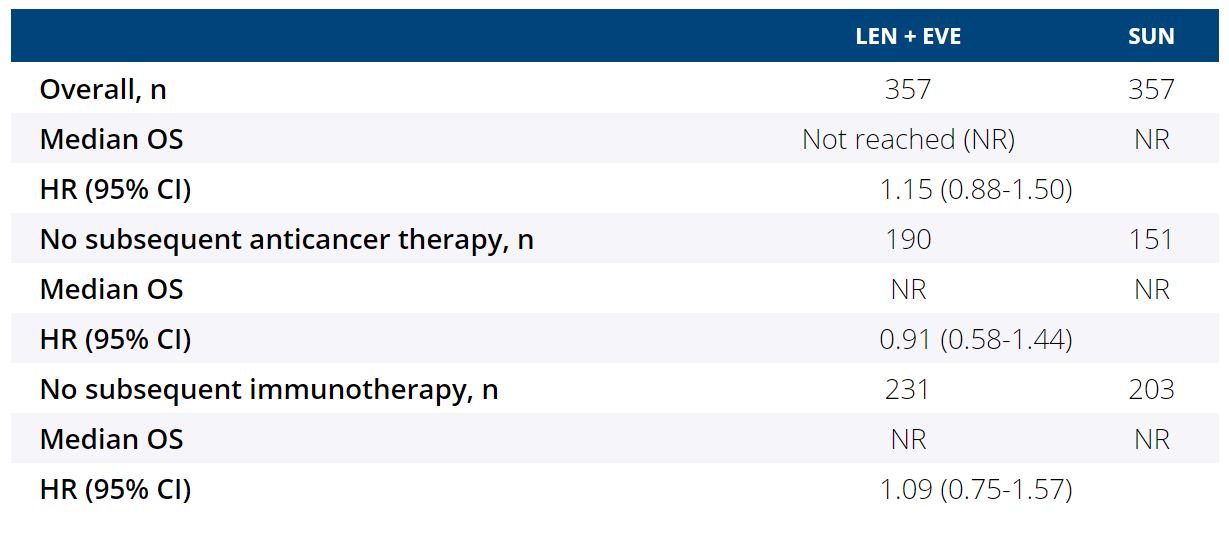
Background:The multicenter, open-label, randomized, phase 3 CLEAR study showed that LEN + EVE had a significant PFS benefit (HR 0.65, 95% CI 0.53-0.80, P<0.001) and improved objective response rate (relative risk 1.48, 95% CI 1.26-1.74) vs SUN in the first-line treatment of patients (pts) with advanced RCC. The difference in overall survival (OS) for LEN + EVE vs SUN was not statistically significant (HR 1.15, 95% CI 0.88-1.50) (Motzer R et al. NEJM. 2021). Post hoc subgroup analyses were performed to assess the impact of subsequent therapy on OS.
Methods:Pts in the CLEAR study were randomly assigned (1:1:1) to 1 of 3 treatment arms, including LEN 18 mg + EVE 5 mg once daily (QD) and SUN 50 mg QD (4 weeks on then 2 weeks off). These post hoc analyses examined OS by subsequent systemic anticancer medication in the LEN + EVE and SUN arms. Hazard ratios (HR; LEN + EVE vs SUN) were based on stratified (geographic region and MSKCC prognostic risk groups) Cox proportional hazards model.
Results:Among 1069 pts with advanced RCC randomized in the CLEAR study, 714 pts were randomly assigned to the LEN + EVE and SUN arms (N=357/each). The median duration of survival follow-up was 27 months in the LEN + EVE arm and 26 months in the SUN arm. Given the shorter median duration of study treatment with SUN (7.8 months) vs LEN + EVE (11.0 months), more pts in the SUN arm received subsequent anticancer therapy during survival follow-up (LEN + EVE, n=167; SUN, n=206). Among pts who received subsequent therapy, pts in the LEN + EVE arm had a longer median time from randomization to initiation of subsequent therapy vs those in the SUN arm (8.0 vs 6.6 months, respectively). OS for the overall population, for pts with no subsequent anticancer therapy, and for pts with no subsequent immunotherapy is shown in the table. In the US population subgroup (LEN + EVE, n=62; SUN, n=61) of the CLEAR study, in which a similar number of pts received subsequent systemic anticancer therapies in the LEN + EVE vs SUN arms (62.9% vs 65.6%, respectively), OS was comparable among the 2 arms (HR 0.95, 95% CI 0.51-1.76). Overall, the safety profile was consistent with the known safety profiles of LEN + EVE and SUN. In both arms, most treatment-emergent deaths were due to progressive disease; there were few treatment-related deaths (<1%, per arm) and no clustering of events.
Conclusions:In the CLEAR study, LEN + EVE met the primary endpoint of a significant benefit in PFS vs SUN. The results of these exploratory analyses suggest that subsequent systemic anticancer therapy affected the OS outcome results for LEN + EVE vs SUN in the CLEAR study. Clinical trial information: NCT02811861
Clinical trial information:NCT02811861
Research Funding: Eisai Inc., Woodcliff Lake, NJ, USA, and Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA 

ABSTRACT 4578:
ASCO Link – https://meetinglibrary.asco.org/record/197514/abstract

Efficacy outcomes of nivolumab + cabozantinib versus pembrolizumab + axitinib in patients with advanced renal cell carcinoma (aRCC): Matching-adjusted indirect comparison (MAIC).
Bradley Alexander McGregor, Daniel M. Geynisman, Mauricio Burotto, Camillo Porta, Cristina Suarez Rodriguez, Maria Teresa Bourlon, Pedro C. Barata, Shuchi Gulati, Brian Stwalley, Viviana Del Tejo, Ella X. Du, Aozhou Wu, Andi Chin, Keith A. Betts, Stephen Huo, Toni K. Choueiri; Dana-Farber Cancer Institute, Boston, MA; Fox Chase Cancer Center, Department of Hematology and Oncology, Philadelphia, PA; Bradford Hill Clinical Research Center, Santiago, Chile; University of Bari ‘A. Moro’ and Policlinico Consorziale di Bari, Bari, Italy; Medical Oncology, Vall d´Hebron Institute of Oncology (VHIO), Hospital Universitari Vall d´Hebron, Vall d´Hebron Barcelona Hospital Campus, Barcelona, Spain; Urologic Oncology Clinic, Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán, Mexico City, DF, Mexico; Tulane Cancer Center, New Orleans, LA; University of Cincinnati Medical Center, Cincinnati, OH; Bristol Myers Squibb, Princeton, NJ; Analysis Group, Inc, Los Angeles, CA; Analysis Group, Inc., Los Angeles, CA; Analysis Group, New York, NY; Dana-Farber Cancer Institute, The Lank Center for Genitourinary Oncology, Boston, MA
Background:Nivolumab in combination with cabozantinib (N+C) has demonstrated significantly improved progression-free survival (PFS), objective response rate (ORR), and overall survival (OS), compared with sunitinib as a first-line (1L) treatment for aRCC in the phase 3 CheckMate (CM) 9ER trial. As there are no head-to-head trials comparing N+C with pembrolizumab in combination with axitinib (P+A), this study compared the efficacy of N+C with P+A as 1L treatment in aRCC.
Methods: An MAIC was conducted using individual patient data on N+C (N = 323) from the CM 9ER trial (median follow-up: 23.5 months) and published data on P+A (N = 432) from the KEYNOTE (KN)-426 trialof P+A (median follow-up: 30.6 months). Individual patients within the CM 9ER trial population were reweighted to match the key patient characteristics published in KN-426 trial, including age, gender, previous nephrectomy, International Metastatic RCC Database Consortium risk score, and sites of metastasis. After weighting, hazards ratios (HR) of PFS, duration of response (DoR), and OS comparing N+C vs. P+A were estimated using weighted Cox proportional hazards models, and ORR was compared using a weighted Wald test. All comparisons were conducted using the corresponding sunitinib arms as an anchor.
Results: After weighting, patient characteristics in the CM 9ER trial were comparable to those in the KN-426 trial. In the weighted population, N+C had a median PFS of 19.3 months (95% CI: 15.2, 22.4) compared to a median PFS of 15.7 months (95% CI: 13.7, 20.6) for P+A. Using sunitinib as an anchor arm, N+C was associated with a 30% reduction in risk of progression or death compared to P+A, (HR: 0.70, 95% CI: 0.53, 0.93; P = 0.015; table). In addition, N+C was associated with numerically, although not statistically, higher improvement in ORR vs sunitinib (difference: 8.4%, 95% CI: -1.7%, 18.4%; P = 0.105) and improved DoR (HR: 0.79; 95% CI: 0.47, 1.31; P = 0.359). Similar OS outcomes were observed for N+C and P+A (HR: 0.99; 95% CI: 0.67, 1.44; P = 0.940).
Conclusions: After adjusting for cross-trial differences, N+C had a more favorable efficacy profile compared to P+A, including statistically significant PFS benefits, numerically improved ORR and DoR, and similar OS.
Research Funding: BMS




Newly Approved Drug Section
ABSTRACT 4567:
ASCO Link – https://meetinglibrary.asco.org/record/197591/abstract

Temporal characteristics of treatment-emergent adverse events and dose modifications with tivozanib and sorafenib in the phase 3 TIVO-3 study of relapsed or refractory mRCC.
Sumanta K. Pal, David F. McDermott, Bernard Escudier, Thomas E. Hutson, Camillo Porta, Elena Verzoni, Michael B. Atkins, Michael N. Needle, Brian I. Rini; Department of Medical Oncology & Therapeutics, City of Hope Comprehensive Cancer Center, Duarte, CA; Beth Israel Deaconess Medical Center, Dana-Farber/Harvard Cancer Center, Boston, MA; Gustave Roussy, Villejuif, France; Texas A&M College of Medicine, Bryan, TX; University of Bari ‘A. Moro’ and Policlinico Consorziale di Bari, Bari, Italy; Fondazione IRCCS Istituto Nazionale Tumori, Milan, Italy; Georgetown Lombardi Comprehensive Cancer Center, Washington, DC; Aveo Oncology, Boston, MA; Vanderbilt-Ingram Cancer Center, Nashville, TN
Background:The randomized phase 3 TIVO-3 study met the primary endpoint of improved PFS with tivozanib (TIVO) vs sorafenib (SOR) in patients with relapsed/refractory mRCC with fewer dose reductions, interruptions and discontinuations despite a longer time on therapy. Greater insight into temporal characteristics of treatment-emergent adverse events (TEAEs) may enable proactive supportive care strategies and improve patient experience.
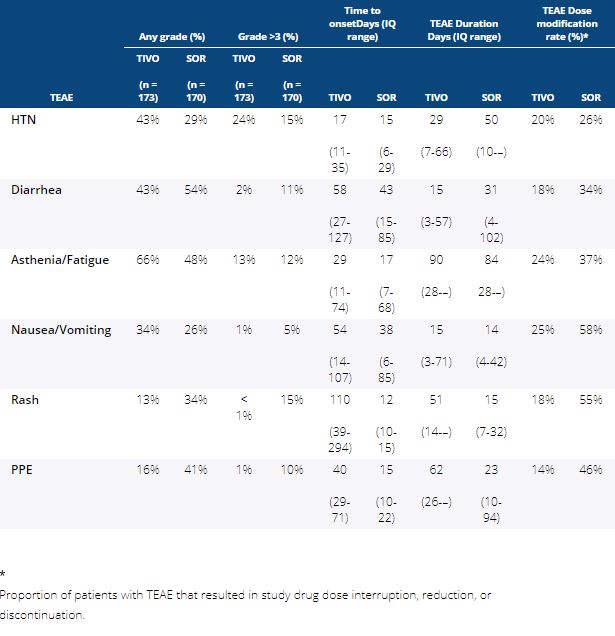
Methods:Updated safety from the previously reported TIVO-3 study with a data cutoff August 15, 2019, was analyzed by treatment arm for time-to-onset (TTO, days [d]) of the most commonly reported TEAEs, and TTO of first dose reduction, interruption, and discontinuation occurring with TIVO and SOR. Duration of TEAE (median d and IQ range), and rate of dose reduction, interruption, or discontinuation due to the TEAE was calculated for each arm.
Results: Patients in the safety analysis randomly assigned to TIVO (n = 173) or SOR (n = 170) received 11.9 and 6.7 cycles, or 336 and 192 mean days of treatment exposure, respectively. Incidence of any Gr, Gr >3, and TTO of any Gr TEAE of special interest occurring with >20% frequency in either arm is shown in Table 1. While TIVO was associated with less Gr>3 diarrhea, rash and PPE and more HTN than SOR, there were few differences in the TTO or duration of these TEAEs. Overall, dose reductions, interruptions, and discontinuations due to TEAEs were less frequent with TIVO than SOR, and TTO of first dose reduction (85 vs 45 d), interruption (81 vs 50 d), and discontinuation (114 vs 49 d) was longer for TIVO than SOR. Among those experiencing the same TEAE in either arm, resulting dose modifications were less frequent with TIVO than SOR.
Conclusions: TIVO-3 demonstrated improved PFS with TIVO compared to SOR in mRCC, with longer duration of TIVO exposure, but fewer all Gr and Gr >3 TEAEs. Temporal characteristics of TEAEs were similar, but time to dose modifications was longer with TIVO than SOR. Among those with the same TEAEs, unmodified treatment was continued more often with TIVO than SOR.

Research Funding: AVEO Oncology
Clinical trial information: NCT02627963.


This material on this page is ©2021 American Society of Clinical Oncology, all rights reserved. For more information, please contact licensing@asco.org.
The post ASCO21 Sessions: RECOMMENDED ABSTRACTS in Renal Cancer first appeared on GUcancers.
]]>The post ASCO21 Kidney Cancer Roundup first appeared on GUcancers.
]]>
Robert A Figlin, MD
Cedars-Sinai Samuel Oschin Comprehensive Cancer Institute, Cedars-Sinai Health System, Los Angeles, CA
The COVID-19 pandemic has exposed fundamental disparities in the provision of health care across our nation and exacerbated the differences in health outcomes associated with race, socioeconomic and other demographic factors. A silver lining however is that pandemic precarity has inspired tremendous scientific collaboration among clinicians, researchers, and key opinion leaders. In this line, this year’s Annual Meeting of the American Society of Clinical Oncology (ASCO21) which was kicked off virtually on June 4 through June 8, not only celebrated latest breakthroughs in cancer research, treatment and patient care, but also focused on health equity in cancer care. ASCO21’s fitting theme – Health equity “doing right by the patients for whom we care” reflected addressing complex forces and systems that have created disparities in cancer care, treatment, and research and identifying ways to ensure that all patients have access to and benefit from the latest cancer advances and high-quality cancer care.Let’s recap the key developments from some of the highest-profile clinical trials presented in the ASCO21 conference.
The results from KEYNOTE-564 trial presented during plenary session of ASCO21 have demonstrated that post-surgery treatment with pembrolizumab extends DFS for clear cell renal cell carcinoma (RCC) patients. This is the first phase III study based on adjuvant immunotherapy to demonstrate an improvement in DFS with a favorable safety profile as compared to S-TRAC and sunitinib. KEYNOTE- 564’s findings support adjuvant pembrolizumab as a potential new standard of care to reduce disease recurrence in patients with fully resected intermediate to high-risk RCC. However, further works remains to be seen especially confirmation of an overall survival benefit, usability in other RCC histologies, and the implications regarding treatment choices in advanced RCC. Additionally the trial included M1 patients post treatment of the oligometastatic disease. This is not a usual cohort of patinets included in classical adjuvant trials and may affect the DFS reported. Currently, there are four IO combinations available for the first-line treatment of metastatic RCC viz. nivolumab plus ipilimumab, pembrolizumab plus axitinib, avelumab plus axitinib, and nivolumab plus cabozantinib.
The latest outcome of CLEAR trial adding lenvatinib plus pembrolizumab combination to the growing armamentarium of first-line treatments for RCC patients. CLEAR met its primary endpoint, with lenvatinib plus pembrolizumab significantly improving progressionfree survival compared to sunitinib. This combination also achieved favorable objective response rate compared to sunitinib with an impressive complete response rate. Lenvatinib + everolimus combination resulted in similar or worse HRQoL and symptom scores compared with patients treated with sunitinib. The additional data builds on previous findings of a phase 3 KEYNOTE-581 trial has shown (Abstract 4502); PD-1 inhibitor pembrolizumab plus lenvatinib improved outcomes versus sutent on a measure of health-related quality of life in first-line renal cell carcinoma (RCC). The additional data showed an improvement of specific health-related quality of life measures including favorable disease-related symptoms scores, as well as better HRQoL and disease-related symptoms scores for physical functioning, fatigue, dyspnea and constipation versus sutent.
Results from KEYNOTE-426’s prespecified final analysis with a 42.8-month median follow-up and a 35.6-month minimum follow-up show that pembrolizumab + axitinib continues to demonstrate superior efficacy over sunitinib with respect to OS, PFS, and objective response rate. KEYNOTE-426 represents the longest follow-up of an anti- PD–1/L1 immunotherapy combined with a VEGF/VEGFR inhibitor for first line RCC and indicate pembrolizumab + axitinib as standard of care for patients with previously untreated advanced clear cell RCC.
Results from CANTATA study shows that the addition of telaglenastat did not improve the efficacy of cabozantinib in mRCC as there was no significant difference in PFS between the arms. While the addition of telaglenastat did not improve outcomes with cabozantinib in this unselected population of patients with clear cell RCC, future studies are warranted to determine the impact of glutaminase inhibition in biomarker-selected patient populations with high dependence on glutamine/ glutaminase, and in combination with other therapeutic partners. A study involving Cabozantinib plus nivolumab (CaboNivo) combination showed that CaboNivo had an acceptable safety profile and showed promising efficacy in metastatic non-clear cell RCC pts with papillary, unclassified, or translocation associated histologies whereas activity in patients with chromophobe RCC was limited.
Latest results from phase 2 study of belzutifan (MK-6482) shown clinical benefit and has a favorable safety profile in patients with VHL disease–associated ccRCC, pNETs, and hemangioblastomas.. In the phase 3 CheckMate (CM) 9ER trial, Nivolumab in combination with cabozantinib (N+C) has demonstrated significantly improved progression-free survival (PFS), objective response rate (ORR), and overall survival (OS), compared with sunitinib as a first-line (1L) treatment for aRCC.In this issue, an exclusive roundtable discussion which I have moderated, participated by three distinguished kidney cancer and genitourinary investigators from across the country focusing on the current challenges in cancer trials post COVID-19 pandemic.
These renowned experts also brainstormed various important topics including cancer therapy delivery in the COVID-19 era, telemedicine, new COVID-19 guidelines, evolving clinical trial design, vaccination efforts in cancer patients, adverse events associated with vaccination, and other newly developed measures and recommendations to manage COVID-19 issues in the provision of cancer care.
Access the Full Roundtable Transcript here.
The post ASCO21 Kidney Cancer Roundup first appeared on GUcancers.
]]>